Case study
Prof G, my neurologist, refuses to tell me if I have brain atrophy or not. He says the neuroradiologists don’t measure it, and even if they did, we can’t use it to make treatment decisions. Do you agree?
Prof G’s opinion
Yes, and no.
Yes, in the sense that an individual patient level brain volume (BVL) measurements are quite wobbly or variable. At the moment, a change in BVL over a 12 month period may simply be within the range of the variability of the measurement and will not indicate real change. I am saying that if you have your BVL measured today and we repeat the measurement tomorrow, there may be a difference of ~0.4% due to the measurement error and not due to true loss in brain tissue. However, very large annual changes, greater than 0.6% or 1.0% per year, are likely to be real. Similarly, a consistent trajectory of BVL over time, say small annual losses over three or four years, is likely to be real and provide a better representation of the pathology. Similarly, a single cross-sectional measurement, say at baseline, is only really meaningful if it is way off the norm, and hence for most pwMS, a single cross-sectional measurement is not that informative.
However, when you use averages across groups in trials, the results are meaningful. So when I say that on average, a group of pwMS treated with alemtuzumab have an annualised BVL of 0.15% and those on fingolimod a rate of 0.40% per annum, it means the group treated with alemtuzumab will do much better on average. Knowing this type of information is hard to ignore when making a decision on which DMT you may want to start. I suspect the neurologist of the patient above may disagree with me on this. What do you think?
An evolving treatment target
I personally think brain atrophy, or BVL, the preferred term for brain atrophy, is an important issue to address when making treatment decisions and should be part of the treatment target. I would be interested to know if this patient had her treatment target discussed with her or not? If not, she should ask her neurologist what it is.
At present most MS services treat to a target of no evident inflammatory disease activity or NEIDA, i.e. being free of relapse and new lesions on MRI. This is why you should have regular MRI scans. The frequency of MRI scans is debatable, with some units doing it every 6 months, some annually and in some situations every two years.
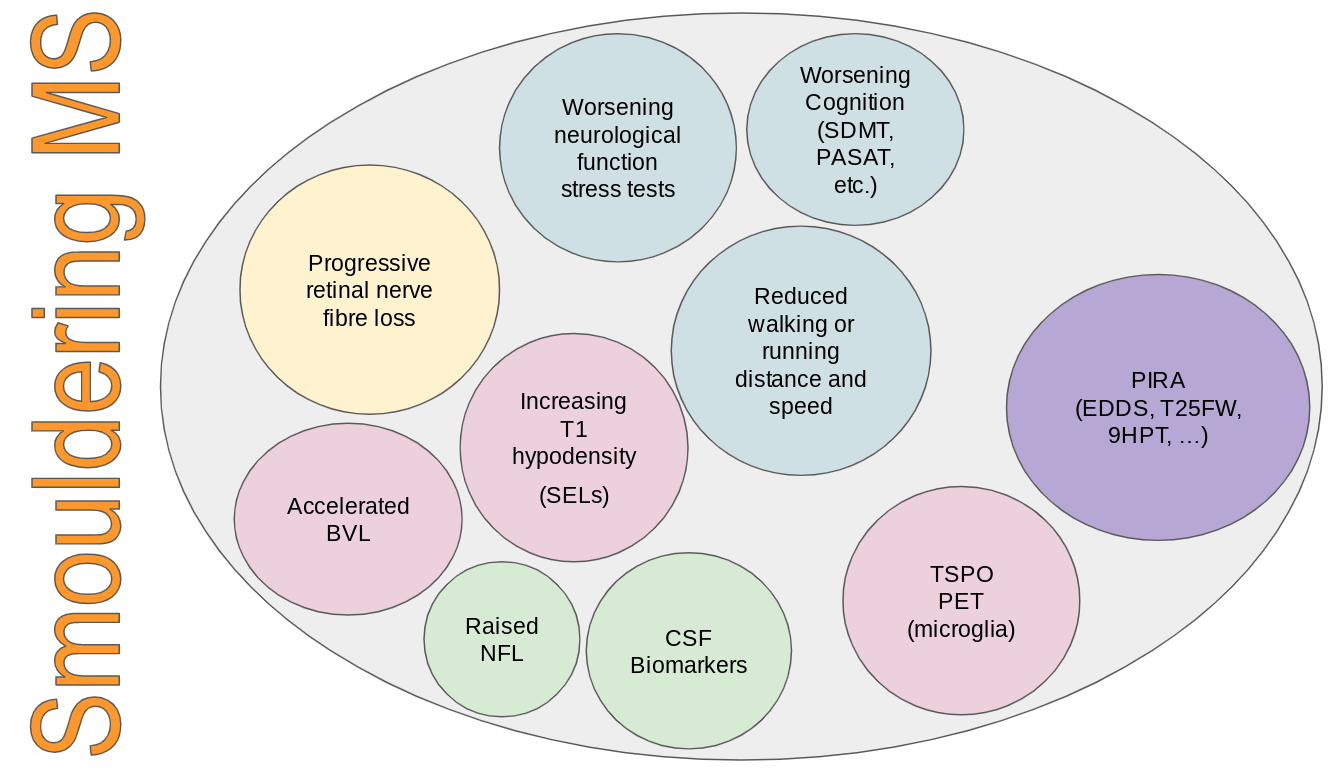
In the recent past, our treatment target was referred to as NEDA (no evident disease activity), which included disability worsening. We know that disability worsening in the absence of inflammatory activity is quite common and occurs in a significant proportion of pwMS on DMTs in clinical trials and in real-life clinical practice. In relapsing patients, this worsening is called PIRA (progression independent of relapses), whereas in progressive patients, i.e. primary or secondary progressive MS, this worsening is what is used to define this stage of the disease. In my opinion, the worsening that occurs in the so-called relapsing and progressive phases of the disease is the same and is the clinically apparent part of what I now refer to as smouldering MS. (please see Newsletter from 27-Jan-2022).
Please note that smouldering MS is much more than PIRA and captures other processes that may or may not be seen clinically, for example, accelerated brain volume loss, progressive retinal nerve fibre thinning, etc.
When we wrote our ‘MS Brain Health: Time Matters’ policy document, we appreciated that the therapeutic target in MS would evolve. Therefore, we made our aim to encourage the widespread adoption of a therapeutic strategy that aims to maximize the lifelong brain health of every person with MS. What we are trying to do here is to get the MS community to think about getting pwMS to old age with a healthy brain as possible. This would then allow them a fair chance of dealing with the ravages of ageing. What protects you from age-related cognitive decline are brain and cognitive reserve. MS reduces both of these, which is why pwMS age earlier than the general population. I recently saw a presentation by a tech company that has developed an algorithm that can estimate your brain age by analysing your brain MRI scan. As expected, the brain age of someone with MS is much older than their biological age. Would you be interested to know your brain age? Or would you find this depressing?
I often get asked that because BVL and ongoing or future BVL predict poor MS outcomes, is there anything we can do about it? Sadly, no. At the moment, there is nothing we can do about brain volume that is already lost. However, if you have ongoing inflammatory MS disease activity suppressing it with effective DMTs may slow down the further loss. What we don’t know is if you are NEIDA on one DMT, say for argument’s sake dimethyl fumarate (DMF), but are still losing brain volume at a rate above normal, for example, 0.6% per annum, would switching you to say alemtuzumab or treating you with AHSCT halt this process. In our centre, we occasionally escalate patients in this situation in the hope that there is ongoing microscopic inflammation and that a higher efficacy DMT will impact BVL. However, I suspect that the processes driving smouldering MS and accelerated BVL need other classes of add-on therapy, hence the need to develop combination therapies in the future and the need to manage MS holistically. The following YouTube lecture explains the underlying principles of going beyond NEIDA as a treatment target in MS and makes a case for the holistic management of MS.
The study below shows how important the NEIDA construct is when we start to think about our treatment target in MS. Don’t we want to suppress MS disease activity as much as possible and protect the end organ (brain and spinal cord) from damage? The analysis of the fingolimod trial data shows that pwMS with the greatest brain volume loss in the first 2-years of the study do much worse in terms of disability progression and further brain volume loss over the next 2 years.
The message is clear to me; if you want to optimise the treatment effect in MS you need to treat to a target beyond NEIDA to protect the end-organ damage and prevent brain volume loss.
I am not giving up on the technology to measure BVL yet. The technology and algorithms to measure it are evolving rapidly, and hopefully, we will have a method that works on the individual patient level very soon. We used to have this same discussion about the routine measurement of T2 and Gd-enhancing lesions in clinical practice, and now most MS centres measure these routinely.
I don’t think I have addressed this patient’s question adequately, but I hope this newsletter provides some context to the thorny issue of BVL in MS and its use in clinical practice. Hopefully, this newsletter will provide a gentle nudge to engage with your annual MRI scan results and to ask your HCP to tell you what has happened to your lesion count over the last 12 months and your BVL over the last few years.
Background: Clinical evidence in patients with relapsing-remitting multiple sclerosis suggests an association between MRI outcome measures and disability progression (DP).
Objectives: Post hoc analysis to investigate the association and potential predictive value of brain volume loss (BVL) with long-term DP in FREEDOMS.
Methods: Patients were categorized into quartiles by SIENA-calculated percent brain volume change from baseline to month (M) 24. Patient characteristics at baseline were determined for each quartile, as were the proportions of patients at M24 and M48 reaching Expanded Disability Status Scale (EDSS) scores of ≥4.0 or ≥6.0 or DP confirmed at 3 months (CDP3) or 6 months (CDP6), and change in EDSS and Multiple Sclerosis Functional Composite.
Results: MS disease activity and severity as well as brain volume at baseline were predictive of subsequent BVL over 24 months. The quartiles of patients with greater BVL at 24 months were at highest risk (odds ratio, p value) for reaching EDSS ≥4 (2.8, p = 0.001) or ≥6 (5.73, p = 0.0005) and experienced more DP at M24 (CDP3 2.13,p = 0.002; CDP6 2.17, p = 0.003) and M48 (CDP3 1.98, p = 0.006; CDP6 1.87, p = 0.018) compared to the quartile of patients with the least amount of BVL.
Conclusions: These findings confirm the clinical relevance of early brain volume changes for long-term DP.
Subscriptions and donations
I am using the paid subscriptions to administer the MS-Selfie Newsletter and associated MS-Selfie microsite that is currently in development. Thank you for being an active paying subscriber; your contribution is much appreciated. At the request of several readers, I have now added the option of making a one-off donation as well. Funds from subscriptions and donations are being used to pay a professional medical writer to curate, rewrite and transfer the contents of the Newsletter onto a companion MS-Selfie microsite, which is being designed and maintained by a freelance web designer.
Thank you.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry or Barts Health NHS Trust. The advice is intended as general advice and should not be interpreted as personal clinical advice. If you have problems, please tell your own healthcare professional who will be able to help you.














Share this post