Case study
Dear Professor Giovannoni
My 17-year-old son has just been diagnosed with multiple sclerosis. As you can imagine, as a parent, I am very concerned about his long-term future. His neurologist has recommended he start fingolimod without much discussion about other treatment options. Is there any advice you can give about the underlying principles of treating MS in young people?
Prof G’s Opinion
As I have said before, how MS is managed and treated worldwide is variable and depends on local practices and guidelines. I suspect your son has been offered fingolimod because fingolimod is the only DMT that has been appropriately tested in the so-called paediatric population, as it was shown to be superior to interferon-beta (Avonex).
I don’t think there is any difference between a 17-year-old (paediatric patient) and an 18+-year-old patient (adult). I would have, therefore, made sure your son was well informed about MS and its management before recommending a treatment. I would almost certainly have recommended multiple treatment options, i.e., I would have offered a choice.
I suggest your son asks the following questions before deciding which treatment to start.
How do I want my MS to be treated? What is the difference between a maintenance/escalation DMT and an IRT (immune reconstitution therapy)?
How can I reduce my chances of adverse events on specific DMTs?
What are the attributes of the specific DMTs or treatment strategies?
Will I be able to become a parent? What about pregnancy and breastfeeding?
Multiple sclerosis (MS) treatment has evolved rapidly with 11 disease-modifying therapy (DMT) classes. I will provide a brief overview of the different classes of DMTs and a classification system for how we think of them and how this fits in a treatment paradigm for using these DMTs effectively and safely. To address the unmet need for treating more advanced MS, including people with MS using wheelchairs, we need a two-pronged approach. A more proactive approach to early and effective treatment of MS with a defined treatment target of no evident inflammatory disease activity (NEIDA), defined as no relapses and no new or enlarging lesions on MRI. In addition, we need to modify our expectation of what can be achieved in more advanced MS and focus on neuronal systems with reserve capacity, for example, the upper limb, speech and swallowing (bulbar function), visual function and cognition. I also appeal to solving the problem of the slow adoption of innovations and the global problem of untreated or undertreated MS.
NEIDA as a treatment target
Relapses and ongoing focal inflammatory activity on MRI (new or enlarging T2-lesions and gadolinium (Gd)-enhancing lesions) are associated with a worse short to intermediate-term prognosis, contradicting natural history studies. These observations have led to the increasing adoption of ‘no evident inflammatory disease activity’ (NEIDA) as a treatment target in MS.
NEIDA is an essential goal for treating individual pwMS and could be used as an outcome in clinical trials. To adopt NEIDA in day-to-day clinical practice, it is advisable to have a ‘rebaseline’ MRI scan after the onset of action of the DMT has been assessed. The timing of the baseline MRI is based on how quickly the DMT works and when you can potentially redose immune reconstitution therapies. Please note that recommendations for immune reconstitution therapies differ from maintenance or continuous therapies. In the case of an IRT, for example, alemtuzumab or cladribine, which are given as short courses of breakthrough disease activity, can be used as an indicator to retreat rather than necessarily switch therapy. Therefore, the rebaselining MRI should be delayed until after the final initial course of therapy, e.g. two years, or close enough to the time when a third or subsequent course can be administered. The question of how many cycles need to be given before considering that a person has failed an IRT remains a moot point until much-needed long-term evidence emerges to guide us.
Compared to IRTs, disease activity on maintenance therapy, provided you have been adherent to treatment, is usually interpreted as a sub-optimal or non-response and a need to escalate or switch therapies to a more effective DMT.
In the past, we used NEDA (no evident disease activity) as a treatment target, which included progression or worsening independent of relapse activity (PIRA) or smouldering MS. PIRA may have little to do with ongoing focal inflammatory activity and may represent the dying back of central nerve fibres as a result of preceding focal inflammatory lesions. Although some of the more effective DMTs may modify this stage of the disease, many neurologists feel uncomfortable switching or stopping a DMT based simply on PIRA or smouldering MS disease activity.
Many HCPs remain sceptical of using NEIDA as a treatment target; they remain critical of the zero-tolerance target. Some are concerned that most pwMS would be on ‘more risky’ high-efficacy therapies. A less aggressive approach uses various scoring systems, allowing minimal disease activity (MEDA). MEDA, however, flies in the face of the science of the focal inflammatory lesion being ‘bad’ and is associated with poor short-, intermediate- and long-term outcomes. If the majority of pwMS end up on the so-called risky high-efficacy therapies because of breakthrough disease activity, then this is what they probably need to have their MS actively treated. Achieving long-term remission, or NEIDA, is a well-established treatment target in other autoimmune diseases, such as rheumatoid arthritis and inflammatory bowel disease. pwMS with MS treated-to-target of NEIDA do better than those with breakthrough disease (at a clinical or subclinical/MRI level). I would, therefore, strongly encourage both pwMS and their HCPs to adopt NEIDA as an initial treatment target for MS. In the future, we must go beyond this to target smouldering MS.
The following figure is an example of implementing a treat-2-target NEIDA strategy. The important take-home message is that the treatment goals in MS have moved and now require setting goals and actively monitoring outcomes to achieve these goals. From now on, there will be a need to regularly update the definition of NEIDA as new technologies become available and are validated as predictors of treatment response. I envisage the definition of NEIDA changing shortly to include more objective metrics, particularly ones measuring end-organ damage, for example, brain volume loss and the inclusion of patient-related outcome measures (PROMS).
Disease-modifying therapies
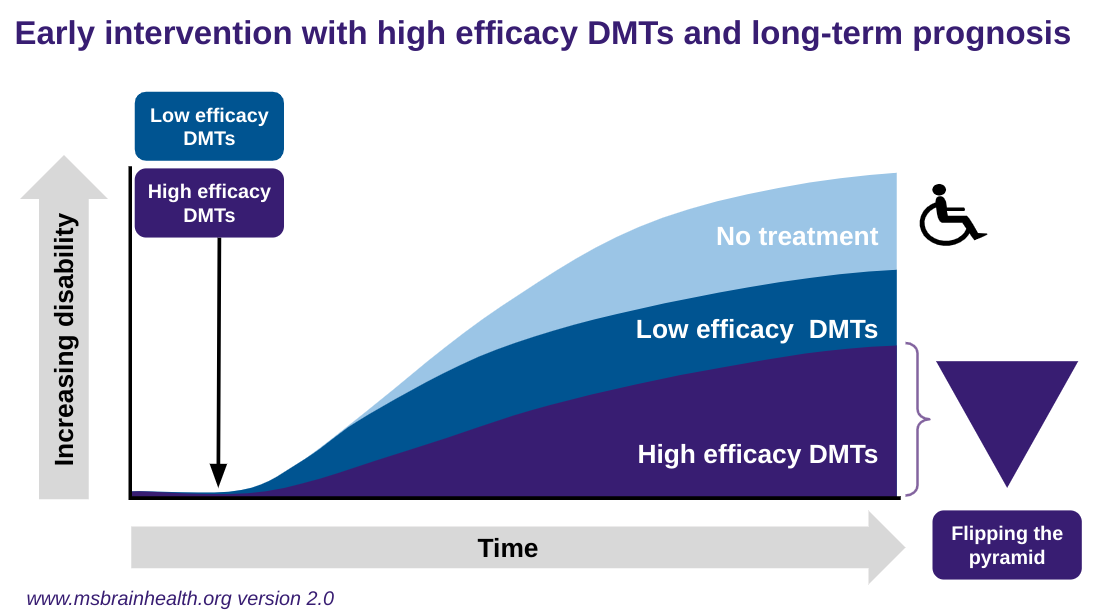
The effectiveness, or relative effectiveness, of individual DMTs becomes less critical in the context of a strategy of treat-2-target of NEIDA. Choosing a DMT with a lower efficacy rate, i.e. a lower NEIDA rate, means that a higher proportion of treated pwMS will need to be switched to higher efficacy therapies over time to achieve NEIDA. In comparison, pwMS choosing high-efficacy therapies will have a greater chance of achieving NEIDA with their initial choice of therapy. Data shows that pwMS treated with the step-care approach do worse on average than those offered rapid escalation or high-efficacy DMTs first-line. The latter of the three approaches is now called flipping the pyramid, i.e. choosing high-efficacy therapies first line. In recent trials of alemtuzumab, ocrelizumab, ofatumumab, and ublituximab, pwMS randomised to 2 years of interferon-beta-1a therapy or teriflunomide had poorer outcomes than those receiving highly-active therapy from the outset. Real-world data from registries also support this; groups of pwMS with delayed access to high-efficacy DMTs do worse than those getting onto high-efficacy treatments early.
About the conventional step care paradigm, pwMS switching horizontally in terms of efficacy from interferon-beta to glatiramer acetate, or vice-versa, i.e. from one moderate efficacy DMT to another moderate efficacy DMT, do not do as well as pwMS switching vertically to fingolimod a highly-effective DMTs. Similarly, people with MS escalating to natalizumab, a very-high efficacy DMT, do better than pwMS being escalated to the less effective, but still high-efficacy, DMT fingolimod.
What determines what efficacy level of a DMT is appropriate depends on individual factors, for example, baseline prognostic profile, family planning, local or national treatment guidelines, socioeconomic factors, patient factors in particular comorbidities, cognitive impairment, risk aversion and lifestyle issues. The adoption rate of the rapid escalation or the flipping of the pyramid over the conventional step-care approach is highly variable worldwide and even within countries.
To address the variation in clinical practice and how MS is treated, we launched a policy document, ‘Brain Health: Time Matters in Multiple Sclerosis’, to activate the MS community to be more proactive in managing MS. We need to update this policy document to include the concept of flipping the pyramid.
One development worth highlighting is the classification of DMTs into maintenance and immune reconstitution therapies and those that are immunomodulatory compared to those that result in immunosuppression. In addition, immunosuppressive therapies can be further subdivided into those that cause prolonged compared to short-term immunosuppression, the relative attributes of which are compared in the following table.
Maintenance vs Immune Reconstitution Therapies (IRTs)
There is a divide between the two main treatment philosophies: maintenance-escalation vs. IRTs. By definition, an IRT is given as a short course, i.e. as a one-off treatment in the case of AHSCT or intermittently as in the case of alemtuzumab, cladribine or mitoxantrone. IRTs are not given continuously, and additional therapy courses are only given if there is a recurrence of inflammatory activity. IRTs can induce long-term remission and, in some cases, potentially a cure. One could argue that none of our treatments cure MS, but unless we define what constitutes a cure and look for it, we won’t find it. At the recent ECTRIMS/ACTRIMS meeting in Milan, I noted that several speakers raised the issue of needing a definition of an MS cure. None of them had read our Editorial from 2013 doing just this.
Banwell B, Giovannoni G, Hawkes C, Lublin F. Editors' welcome and a working definition for a multiple sclerosis cure. Mult Scler Relat Disord. 2013 Apr;2(2):65-7.
In comparison, maintenance therapies are, by definition, given continuously without an interruption in dosing. Although maintenance therapies can induce long-term remission, i.e. NEIDA, they cannot, by definition, result in a cure. With maintenance therapies, the recurrence of, or ongoing, inflammatory activity indicates a suboptimal response to treatment and typically results in a treatment switch. Ideally, this switch should be to a more effective class of DMTs, hence the term escalation.
Continuous and intermittent immunosuppression
Another practical way of classifying DMTs is based on whether or not they are immunosuppressive. An immunosuppressive is any DMT that reduces the activation, or effectiveness, of the immune system. From a regulatory perspective, for a drug to be classified as being immunosuppressive, it should (1) cause significant lymphopaenia (low lymphocyte count) or leukopaenia (low white blood cell count), (2) be associated with opportunistic infections, (3) reduce the antibody response to vaccines and (4) and increase the risk of secondary malignancies. The duration and intensity of immunosuppression further determine the risks. For example, short-term or intermittent immunosuppression associated with IRTs front-loads the risks, which are substantially lower once the immune system has reconstituted itself.
In comparison, long-term continuous or persistent immunosuppression, which occurs with some of the maintenance DMTs, accumulates problems over time, particularly opportunistic infections and secondary malignancies. The following table summarises the main attributes of intermittent and persistent immunosuppression. Please note that live vaccines are generally contraindicated in pwMS on immunosuppressive therapies. In comparison, pwMS on IRTs who have reconstituted their immune systems can tolerate and respond to live vaccines. The decision to administer live vaccines in this situation must be balanced against the risks of the vaccine.
Advanced MS
I have had several debates with a colleague about therapeutic targets and whether or not they are achievable. In general, many consider NEIDA an unrealistic treatment target as most of our pwMS who achieve NEIDA still have residual problems. Therein lies the problem. NEIDA refers to no evidence of ongoing inflammation. NEIDA does not refer to the reversal of previous damage, nor does it refer to the ongoing consequences of previous damage. You can only do so much with an anti-inflammatory agent, i.e. switch off inflammation, and even then, most anti-inflammatory agents don't switch off innate immune activation within the CNS, for example, activated microglia, nor do they necessarily purge the nervous system of B-cells, plasmablasts, plasma cells and their oligoclonal immunoglobulin products that are considered to plan a major role in worsening disease or smouldering MS. We can only ask so much of our current DMTs, which is why in the future we will need combination therapy strategies to target other pathways.
I have previously proposed that we consider rejecting the adjective ‘progressive’ to describe the worsening disability in MS. Progression is a misnomer and means improvement. I prefer the term ‘worsening MS’, which captures the associated disability that comes with the later phases of the disease. As a community, we must accept the pathology that drives neuroaxonal loss or neurodegeneration (the pathological substrate that underlies ‘worsening MS’) as being there from the beginning. This means the neurodegenerative phase of MS is present from the beginning, long before pwMS become physically disabled.
In my opinion, MS is one disease and not two or three diseases. Scientific or philosophical arguments do not back up the division of MS into several diseases. The false division of MS into several diseases has become counter-productive to the field of MS. The division of MS into relapsing and progressive forms was Pharma-led to get MS defined as an orphan disease, which allowed interferon-beta-1b to get a license in the United States based on the results of one pivotal phase 3 study.
Following the abovementioned point, the division between SPMS and PPMS is also false. No pathological, genetic, imaging or other data suggests these are different entities. We, therefore, should be doing trials in both populations simultaneously. Another dogma that needs addressing is the proposal that more advanced MS is ‘non-inflammatory’. There are clinical, imaging and pathological data that show inflammation still plays a big, and probably a major, role in advanced worsening or smouldering MS. Therefore, not targeting more advanced MS with an anti-inflammatory is folly and may explain why so few single therapy neuroprotective trials have been unsuccessful. The fact that ocrelizumab has been licensed for active primary progressive MS and siponimod for active secondary progressive MS implies they will form the platform for future add-on trials.
It is essential to acknowledge that reserve capacity in particular neuronal systems plays an integral part in how MS worsens. Neuronal systems with reserve capacity are more likely to recover function and hence show a treatment effect than neuronal systems in which reserve capacity is exhausted. In the latter systems, showing a treatment effect will take longer; I refer to this as therapeutic lag. These observations are explained by the length-dependent axonopathy hypothesis of worsening MS, i.e. progressive MS manifests initially in pathways with the longest axons (bladder and lower limb motor function). Therefore, we should focus more on the arm-and-hand function as a primary outcome in pwMS who have already lost too much function in their lower limbs (EDSS>=6.0). Once someone with MS has lost lower limb function and become a wheelchair user, they still have neuronal systems that are potentially modifiable, for example, upper limb, speech, swallowing, visual and cognitive function. There is an extensive evidence base showing that several licensed DMTs can slow the worsening of upper limb function despite subjects having advanced MS. I feel very strongly about this point and have argued for future trials in advanced MS to include wheelchair users with a focus on upper limb function as the primary outcome measure. Their arm and hand function keeps pwMS independent and functioning in society.
Another trend beginning to emerge is the need to use combination therapies in advanced MS. This does not necessarily exclude using two classes of anti-inflammatory therapies, provided their mechanisms of action are complementary. There are good arguments for combining agents that target innate immune mechanisms - for example, drugs that target activated glia - with a classic anti-inflammatory targeting adaptive immune mechanisms that are presumably MS-specific, for example, anti-B and anti-T cell-driven autoimmunity. However, a combination of an anti-inflammatory targeting adaptive immune responses and a neuroprotective or remyelination therapy makes the most sense.
We also need to tackle ageing and its impact on worsening MS. The evidence that early, or premature, ageing from the reduced brain and cognitive reserve drives the worsening of MS in older pwMS is, in my opinion, beyond doubt. What we need is some way of dissecting premature ageing from MS-specific mechanisms. Another issue with ageing is the emergence of comorbidities as a driver of worsening MS, mainly smoking, hypertension, hypercholesterolaemia, metabolic syndrome, diabetes, social determinants of health and a sedentary lifestyle.
Despite the promise of treatments to modify the course of more advanced MS, we need to manage expectations. PwMS expect an effective treatment to restore function or return them to normal. This is not going to happen. The best we can expect is to slow down the rate of worsening disability, or flat-line disability progression, with a combination of anti-inflammatory and neuroprotective strategies. Although it is plausible that neuronal systems and pathways with reserve capacity can show improvement in function, it is unlikely sufficient to make predictions on future outcomes and falsely raise the hopes of pwMS. To get substantial and meaningful improvements in disability, we will need new treatments, including remyelination therapies and therapies that promote neuronal growth (axonal sprouting), new nerve connections (synaptogenesis) and plasticity mechanisms to restore function.
For more information on advanced MS, I would suggest reading the MS-Selfie Newsletter ‘Getting Worse’ (2 Jul 2021).
Conclusions
Over the last 30 years, the treatment and management of MS has been transformed. Who would have thought we would target NEIDA, and potentially end-organ damage, as a realistic treatment target for pwMS and define a potential MS cure? The MS community is now considering slowing down disability worsening in the more advanced stages of the disease, wheelchair users, and exploring add-on neuroprotective and neurorestorative therapies. Despite all the hype of the future, the biggest single problem we in the MS community face is slow and, in some circumstances, meagre adoption rates of innovations. Far too many pwMS remain untreated or under-treated with DMTs. In many parts of the world, this is simply due to economic factors and the unaffordability of high-cost innovator drugs. We must think of creative ways to get people living in resource-poor environments to access effective DMTs. Including three DMTs in the WHO’s essential medicines list may change this. However, this may involve compassionate-use DMT access programmes or establishing an evidence-based list of essential low-cost, off-label DMTs.
Subscriptions and donations
MS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, weekly off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite, as I don’t have time to do it myself. You must be a paying subscriber to ask questions unrelated to the Newsletters or Podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).
Important Links
General Disclaimer
Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Queen Mary University of London or Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your own healthcare professional, who will be able to help you.















Share this post