I have just arrived back from ECTRIMS 2022 in Amsterdam and have reflected on a week of meetings. I had little time to view abstracts and/or attend live sessions. I am hoping to catch up with missed content using the on-demand service. Despite this, I managed to get an overview of what is important from discussions with friends and colleagues.
1. EBV
EBV remains, in my opinion, on top of the pops. The good news is that almost all of the pharmaceutical companies I spoke to are now including EBV in their long-term thinking. Some companies are even planning trials targeting EBV directly as a therapeutic target; others are doing it without quite realising they are doing it, for example, with their Bruton Tyrosine Kinase (BTK) inhibitors (BTKi). EBV uses BTK to maintain a pro-survival signal for memory B-cells to stay alive. Ibrutinib, a first-generation BTKi, has been shown to inhibit EBV-induced B-cell proliferation and works well against EBV-associated lymphomas, including CNS-lymphomas. The latter is why Professor David Baker and I tried unsuccessfully to get a trial of ibrutinib started in MS about seven years ago.
The EBV late-breaker on the immune response to EBV in people with MS has already been published, and I have commented on this. If you are interested in my interpretation of this research, please read my previous MS-Selfie Newsletter (More evidence that EBV causes MS, 6-Sept-2022).
2. Bruton Tyrosine Kinase (BTK) inhibitors (BTKi)
It was clear at ECTRIMS 2022 that the BTKi’s are the next big thing. There are four phase 3 trial programmes underway (Evobrutinib, Merck; Tolebrutinib, Sanofi; Fenebrutinib, Roche; Remibrutinib, Novartis) with a fifth (Orelabrutinib, Biogen) about to start and a sixth product (GB7208, Gossamer) in the wings waiting to join the rush.
There was a lot of discussion at the meeting about the emerging liver toxicity signal identified with this class of agents. Whether this is a problem for one or two compounds or the whole class is too early to tell. However, the pharmacovigilance monitoring that goes with agents with a liver toxicity signal will add friction to the prescribing of this class unless it can be derisked or made simple, for example, with a home or self-monitoring solution. I suspect outside of the MS market, there is a big need for home blood monitoring for pharmacovigilance purposes using connected devices that report data directly to HCPs via the cloud. Are there any entrepreneurs out there who are or can develop such devices? We are ready to adopt them into clinical practice.
The first COVID-19 vaccine data was presented in relation to evobrutinib at the meeting, and to my surprise, antibody and by implication T-cell responses were not blunted. BTK is critical in driving antigen-specific B-cell responses, i.e. people who are born without BTK have a condition called Bruton’s agammaglobulinemia, i.e. they don’t have circulating immunoglobulin. So the fact that evobrutinib-treated patients with MS make antibodies to COVID-19 vaccines tells us that evobrutinib cannot be a very potent inhibitor of the enzyme or is not penetrating deep tissue compartments such as the lymph nodes to inhibit germinal centre function. This also questions whether or not CNS penetration will be sufficient to inhibit CNS resident B-cells and plasmablasts. This data indicates that evobrutinib is unlikely to have a potent immunosuppressive profile. It will be interesting to see what happens with the other BTKi’s in development.
3. Radiologically-isolated syndrome
I have been running a campaign to redefine MS as a biological rather than a clinico-radiological disease for several years; this is my #MS_is_1_not_2_or_3_diseases campaign. Part of redefining MS as a biological disease is including asymptomatic MS or RIS (radiologically-isolated syndrome) as part of the diagnostic criteria. The good news is that the first disease-modifying therapy (DMT) trial in RIS was reported as a late-breaker at ECTRIMS, and it came as no surprise that it was a positive trial. Yes, people with RIS randomised to dimethyl fumarate (DMF) had over 80% less chance of having a first clinical event and being diagnosed with clinically-isolated syndrome (CIS) or MS over two years.
The reason I am not surprised by this result is that my proposed definition of MS already includes RIS as MS and if DMF works in MS of course, it is going to work in RIS. This trial result means the McDonald criteria committee will now have to include a new diagnostic category of MS, i.e. asymptomatic MS and maybe even a category for prodromal MS.
4. Ocrelizumab vs. Rituximab
For some time, I have argued against the wide adoption of rituximab and extended interval dosing for ocrelizumab in high-income countries. I have based my argument on the evidence that subjects in the pivotal phase 3 ocrelizumab trial programme with the highest exposure to ocrelizumab did better in relation to disability progression than subjects exposed to lower levels of ocrelizumab. These observations underpin the current trial programme looking at high-dose ocrelizumab vs low-dose ocrelizumab in both relapsing and primary progressive MS.
It is, therefore, unsurprising to me that ocrelizumab was more efficacious than rituximab in this registry study below (Roos et al.). This is not a randomised study but depends on matching subjects at baseline using propensity matching/ It, therefore, requires a randomised study to confirm these results. There are several head-2-head rituximab vs ocrelizumab trials underway to test the hypothesis that rituximab is non-inferior to ocrelizumab. I predict that these trials will show the opposite that rituximab is inferior to ocrelizumab, particularly in relation to disability progression and brain volume loss. Based on this data, we have to assume that rituximab is likely inferior to ocrelizumab when ocrelizumab is dosed at 600mg every six months and that we should, where possible, continue to prescribe the licensed product.
I suggest you read the following MS-Selfie newsletters that cover this issue in more detail:
Case study: are we ready for adaptive ocrelizumab dosing? (2-Jan-2022)
Case study: ocrelizumab or ofatumumab, which would you choose? (27-Aug-2021)
5. AHSCT vs. Natalizumab
The proponents of the AHSCT for all things MS will be surprised that in patients with more advanced MS, i.e. those with progressive MS, AHSCT was not more effective than natalizumab.
The study below of patients with secondary or primary progressive MS from six AHSCT MS centres in Ottawa, Uppsala, Sheffield, Bergen, Sydney and Melbourne were combined and compared with patients from MSBase (Kalincik, et al). Patients were included if they received AHSCT or commenced natalizumab (baseline) once they had been classified as having progressive MS. This study uses the same propensity scoring technique as the rituximab vs. ocrelizumab study above. It is also clear from the study that AHSCT is associated with higher life-threatening adverse events.
Based on this analysis, it will be hard to justify referring patients with progressive MS for AHSCT. The caveat in the UK is that natalizumab is not licensed for progressive MS. It is a great pity that when Biogen did the ASCEND trial of natalizumab in SPMS they didn’t extend the trial to 3 years in duration. If they did, we would certainly have natalizumab as a treatment option for SPMS.
For further information on this topic, please read my MS-Selfie newsletter, “Why is natalizumab not licensed to treat progressive MS? (17-Jun-2022)”.
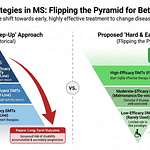
6. Flipping the pyramid and smouldering MS
A major theme at this year’s ECTRIMS was the push for flipping the pyramid at baseline, i.e. offering highly effective therapies as first-line therapies that results in better overall outcomes.
Despite the evidence favouring flipping the pyramid, there has been a slow adoption of this treatment strategy, with the undertreatment of MS emerging as a significant problem. At the same time, a philosophical approach using deductive reasoning indicates that the treatment target in MS needs to go beyond focal inflammation, i.e. relapses and focal MRI activity, and target end-organ damage and the processes driving smouldering MS. There is evidence to argue that the ‘real MS’ is driven primarily by smouldering disease and that focal inflammation is in response to these smouldering processes and not the cause of these processes.
In natural history studies and clinical trials, relapses and focal MRI activity in MS patients on placebo or on disease-modifying therapies (DMTs) are poor predictors of long-term disease evolution and are dissociated from disability outcomes. Most of the accumulation of disability in MS occurs independently of relapse and MRI activity from early on in the course of the disease. Therefore targeting a state of ‘no evident inflammatory disease activity’ (NEIDA) cannot sufficiently prevent disability accumulation in MS, meaning that treatment should focus on pathological processes contributing to the slow loss of neurological function. This approach needs to be complemented with a holistic approach to managing other systemic disease processes that have been shown to worsen MS outcomes.
Saying this, there is, however, a need to manage unrealistic expectations of what can be achieved in more advanced MS, focusing on neuronal systems with reserve capacity, for example, the upper limb, bulbar (speech and swallowing), cognition and visual function.
The following is a practice video of the presentation I gave at ECTRIMS on this topic.
7. Vitamin D
Another highlight was the poster on the PrevANZ results that showed that vitamin D supplements do not prevent the development of MS in patients with CIS (clinically-isolated syndrome) (Butzkueven et al.). People with CIS taking part in the trial were randomised to one of three different daily doses of vitamin D (1000 IU (international units), 5000 IU or 10000 IU), or a placebo (no vitamin D). Vitamin D was used as a standalone therapy - study subjects were not on other disease-modifying therapies for CIS. Subjects were then followed for 48 weeks to determine whether they went on to develop MS. Doses of up to 10,000 international units per day did not reduce MS activity compared to those who did not take vitamin D.
This supports the hypothesis that low vD levels in pwMS are due to vD consumption and vD is not a DMT. The latter is reverse causation, i.e. MS results in low vD levels rather than the other way around. This study, however, does not answer the question of whether or not vD supplementation can prevent MS. As you are aware, people with CIS already have MS, i.e. biological disease, and taking vD after developing MS may be too late in the causal pathway to impact the disease trajectory.
Should pwMS stop taking vD supplements? I would say no, as low vD levels are a risk factor for developing osteopaenia and osteoporosis, which is a major problem in pwMS. So vD supplementation and ensuring you are vD replete is about bone health rather than treating your MS.
Some people will criticise this study for not studying even higher doses of vD. However, the evidence that very high doses of vD make any difference to MS outcomes is very poor and based on observational data, which, unfortunately, is biased. This is why I don’t recommend very high or ultra-high vD supplements in patients with MS.
For more information on vitamin D supplements in pwMS I would suggest reading my MS-Selfie newsletter “Vitamin D supplements: what dose? (7-Sept-2021)”.
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry nor Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your healthcare professional, who will be able to help you.