Are you reporting all your relapses? If you don’t, it could have consequences.

Case study
The problem of not reporting and documenting relapses is an issue for people living with MS (pwMS). I saw a patient nine years ago who probably had MS for at least six years. He was diagnosed with a clinically-isolated syndrome (CIS) after presenting with the MS hug (tight constriction band around the chest), loss of feeling from the waist down, and weakness in the legs. He had an abnormal brain MRI and a positive spinal fluid analysis when he presented. After a CIS diagnosis, his neurologist decided that watchful waiting was the best therapeutic strategy; he was told that not all people with CIS go on to develop MS and that the treatments available were not that effective and came with potentially serious adverse events.
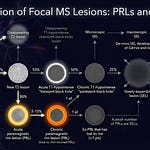
Over the next six years, this patient probably had mild attacks, with patches of sensory loss and tingling in the arms and legs. Although he was seen at the clinic annually, these symptoms were dismissed as possibly being due to minor relapses, and because they were not documented, his diagnosis remained that of CIS. Tragically repeat MRI studies were not done to determine the extent of the subclinical disease activity.
He was referred for a second opinion after he noticed progressive weakness in the legs over the last 12 months.
Prof G’s opinion
When I saw him in the clinic, he used a walking stick. He said that about a year ago, he had had leg weakness that came on quite suddenly over a few weeks, suggesting a possible relapse, but since then, he has progressed. He was treated with oral steroids, which made little difference to his functioning.
In addition to weakness, he had bladder problems, chronic constipation and sexual dysfunction. He was depressed and anxious about losing his job. At the time, he was having increasing difficulty at work, mainly due to memory problems. He tells me he forgets to complete tasks. His MRI showed a massive lesion load, gross brain atrophy and four gadolinium-enhancing lesions.
Although he has focal inflammation on his MRI and a recent history of a disabling relapse, I suspect he probably has entered the so-called secondary progressive phase of the disease. To diagnose SPMS, you must have a history of progressive MS for at least six months. When I spoke to him about his cognitive problems, they had worsened for at least two years. Despite this, I gave him the benefit of the doubt and offered him a DMT with a recommendation for going onto natalizumab, a highly-active DMT.
This gentleman has only had one documented relapse in the last 12 months. The other relapse that ‘qualified’ him for treatment was subjective, i.e. a sensory attack with numbness and pins and needles in the feet associated with unsteadiness of gait about eight months earlier. During this relapse, he struggled to climb stairs and needed a handrail for support.
Many of my colleagues would not have classified this as a relapse as he was not examined during the attack, and hence it was not documented. Some neurologists would have said that as this was not a disabling attack, he could still function “normally” through it, nor did he require steroids or hospital admission. This is not just semantics; in the NHS, we need two documented disabling relapses in 12 months to be deemed eligible for natalizumab, and we need an active MRI scan at baseline. Despite this, it is my policy to give the patient the benefit of the doubt, and any relapse affecting normal activities of daily living is disabling to me. For a Londoner using the underground, not being able to walk down the escalators is disabling.
The good news is this patient has been NEIDA (no evident inflammatory disease activity) on natalizumab for eight years and is still on the treatment. His EDSS has worsened, and he is now EDSS 6.5, but he has maintained upper limb function. Although he stopped working, he now does unpaid charitable activities.
I can only wonder what would have happened if this young gentleman (only 40 years of age) had been started on treatment at the CIS stage and had his MS actively monitored with relatively frequent MRI studies. Would the DMT have prevented him from developing cognitive impairment? Would he be fully mobile? Would we have prevented his brain from shrinking? Would he have normal bladder and bowel function? Would his sex life be normal? Would he still be employed?
Based on phase 3 trial data, extension studies, and more recent real-life registry data, early access to DMTs and particularly early access to highly-effective treatments delays disability progression and the time to diagnosis of SPMS. Once you acquire end-organ damage, the benefits of treatment are much reduced. It is my experience that people with more advanced MS starting high-efficacy DMTs still benefit, i.e. the DMT can suppress relapses and MRI activity, but progression continues for a few years before plateauing out. It is also unusual to see much recovery, particularly in older patients and when there is evidence of end-organ damage on MRI (brain volume loss or black holes on T1-weighted MRI).
The plateauing-out observation is due to therapeutic lag, i.e. progression occurring now and in the next 12-24 months is due to damage acquired in the past, and the benefits of starting a highly-effective treatment will only be seen in the future. I have discussed this issue in detail in a previous Newsletter called ‘Getting worse’ (2-July-2021).
I know this patient’s story is historical and that most neurologists now treat patients with CIS. However, we still have problems with the NHS in that only the old injectable therapies (interferon-beta and glatiramer acetate) are available for CIS. Another strategy that can be adopted is the strict application of the new McDonald criteria that allows the detection of local synthesis of oligoclonal IgG bands in the spinal fluid to count as dissemination in time. This allows the earlier diagnosis of MS and at least opens up other treatment options, i.e. dimethyl fumarate, teriflunomide, ponesimod and anti-CD20 therapies (ocrelizumab and ofatumumab). Sadly, the other high-efficacy therapies can only be offered for patients with rapidly evolving severe MS. This is why I am lobbying Merck and Biogen, the manufacturers of cladribine and natalizumab, to apply to the MHRA for label changes to allow us to use these agents first-line in patients with active MS. Do you agree with me?
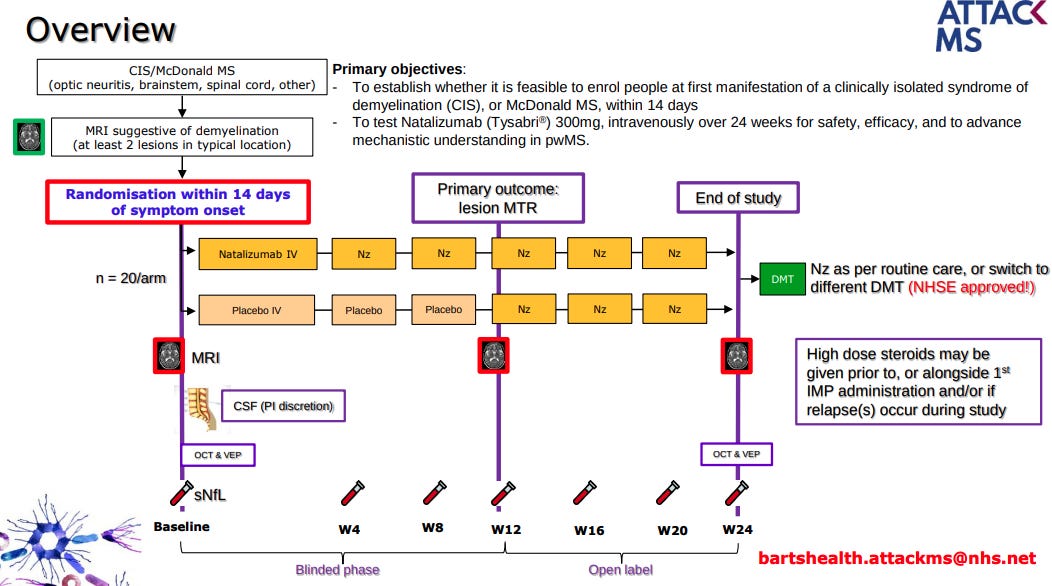
We are so concerned about delayed access to highly effective treatment in MS that we have started the Attack-MS trial, which is testing whether or not treating MS as soon as possible with natalizumab (within 14 days of symptom onset) improves outcomes compared to delayed access to natalizumab (3 months). This trial aims to approach the treatment of MS like we treat stroke. In stroke, minutes and hours make a difference. In MS, we think days and weeks matter. Professor Klaus Schmierer is leading this study, and the following slides explain the study design.
If you or a family member presented with a first attack of MS, would you participate in this study?
We need your help; please spread the message about this study locally. It will be a complex study to recruit for as it is challenging therapeutic inertia and if positive, will change the MS treatment paradigm.
Apart from effective early treatment and flipping the pyramid (high-efficacy DMTs first-line), the other learning point in this Newsletter is not to ignore symptoms of potential disease activity. Don’t ignore new symptoms. New symptoms may mean you are having a relapse, and your current therapy is failing you. If in doubt, contact your HCP for a relapse assessment.
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription, please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry nor Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your healthcare professional, who will be able to help you.