I did an interview yesterday for a documentary about the causal role of EBV in multiple sclerosis. Despite writing and talking about this issue extensively, I was asked what pivotal questions/experiments need to be answered/done, who will be doing them, and how much the research will cost. I promised to put my thoughts down in writing. I apologise for repeating myself, but sometimes you must repeat yourself to get the message across.
Q1: Does EBV vaccination to prevent infectious mononucleosis (IM) prevent or reduce the incidence of multiple sclerosis and other EBV-related disorders?
The vaccine will be developed and tested by the big vaccine companies ($100M+). However, large international registries will be needed to answer whether the incidence of MS decreases after the vaccine's introduction. The latter will need national funding ($2M+). In addition, we need an international policy initiative and public education programme to ensure governments implement EBV vaccination at a national level and that the general public accepts the need for vaccination ($2M+).
Q2: Does treating EBV-associated infectious mononucleosis (IM) with antivirals reduce the incidence of multiple sclerosis and EBV-related disorders?
There are two prongs to this question. Firstly, licensed antivirals are repurposed to treat EBV-associated IM ($2M+). This can potentially be done by academics; we plan to use famciclovir. We can only apply for funding for this study once we have shown an effect of famciclovir on the salivary shedding of EBV. The latter study is almost completed.
The second prong is the development and testing of new antivirals targeting EBV. This will need to be done by pharmaceutical companies. I am in the process of trying to get several pharmaceutical companies to test their antivirals as a treatment for acute IM ($100M+ per drug). The cost of any new drug development programme is vast, which is why this has to be done by the pharmaceutical industry.
Only once a treatment for IM is widely available and adopted by healthcare systems can we look for the impact this is having on the incidence of MS in the general population. This, again, will need national registries, which will best be done in the Nordic countries where these registries already exist.
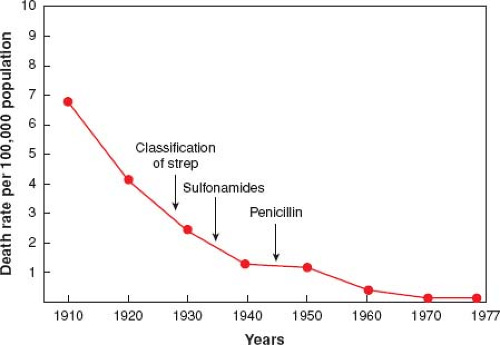
An analogy to the IM treatment paradigm is rheumatic fever, an autoimmune heart disease triggered and driven by streptococcal infections, typically recurrent tonsillitis or pharyngitis. However, once antibiotics emerged, it became obvious that the incidence of rheumatic fever was decreasing (see figure below), which led to successful trials of antibiotic prophylaxis that were shown to prevent recurrences or relapses of rheumatic fever.
Q3: Does treating MS with anti-EBV therapies change the natural history of MS, i.e. are antiviral DMTs?
This question can be answered in several ways. Firstly, by studying existing DMTs and showing that their efficacy is closely associated with anti-EBV effects. This is easier said than done. We are unsure if we need to target EBV in the peripheral blood, secondary lymphoid organs, saliva or central nervous system. Nor do we have validated biomarkers to measure EBV biology in these hard-to-get-at-compartments. Another question is whether antivirals that only target active or lytic EBV infection will be sufficient or whether we will need to use antivirals that target both latent and lytic infection, such as EBNA-1 inhibitors.
I am working on the premise that we will need to target both the central nervous system and the peripheral compartments and that we will also need antivirals to target both latent and lytic cycles of the virus. This is why several pharmaceutical companies are developing immunotherapies to target EBV-infected cells. Immunotherapy is based on the principle that T-cells will traffick to these hard-to-get-at-compartments and find and kill EBV-infected cells.
Immunotherapy can be via the transfer of autologous or allogeneic EBV-specific T-cells, CAR T-cells targeting B-cells or therapeutic EBV vaccines, to name a few approaches.
However, the assumption that we may need to target latent EBV infection and the CNS shouldn’t stop us from doing trials of antivirals that only target the peripheral compartment and lytic infection. The results of these studies will help answer the question of how EBV causes MS. This is why it may be worth creating an adaptive trial platform so many arms can be tested at once, and new arms added as new anti-EBV strategies come along.
In my interview, I was asked which drug I would take forward now. Based on the literature and data available, I replied that I would do a trial of HAART (highly active antiretrovirals), and I would start it ASAP. The price tag for a properly-powered phase 2 trial would be in the order of $5-7M on the condition we get one of the pharma companies who make HIV drugs to give us the drug for free.
Why HAART? There is much anecdotal evidence that people with MS who start HAART do well. People with HIV have a reduced risk of getting MS. We think this is not due to HIV infection but the HAART they take to suppress HIV. Many HIV drugs suppress EBV and may also work against HERVS (human endogenous retroviruses), which may play a role in MS downstream of EBV. Interestingly, EBV is one of the stimuli that transactivates HERVs, which may mediate the damage in MS. The EBV-HERV link is often referred to as the dual viral hypothesis.
Does $5-7 million sound expensive? Would you support such a study? Would you participate in such a study?
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription, please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry nor Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your healthcare professional, who will be able to help you.