Important notice
This newsletter comes with a health warning as it discusses the issue of death in people with multiple sclerosis. I believe you should be aware of this information as it helps balance the risks and benefits of treating MS and the dangers of untreated MS. It also shows how DMTs are changing the long-term outcome of MS. I think the overall message should be a positive one in that in the modern era, if we treat MS early and effectively we will prevent or delay the downstream consequences of MS.
Case study
Recently in one of my clinics, a young woman became very tearful and upset after her partner had been diagnosed with MS. She expressed being very anxious about her partner potentially dying in his forties from MS. This woman has been using Dr Google to find out about MS and come across the life story of Jacqueline du Pré who tragically died with MS at the age of 42. This young woman had watched several YouTube clips and the documentary on Jacqueline du Pré. She had become fixated on the idea that MS is a fatal disease and kills you in your early forties.

Prof G’s response
This person’s experience is one of the downsides of Google and is likely to be a major problem with AI chatbots such as ChatGPT; despite my early enthusiasm for AI and its role in the management of MS (see ‘Happy New Year: broken promises’; 1-Jan-2023)
Jacqueline du Pré is not representative of MS nor its prognosis. Du Pré developed and died from MS in the pre-DMT era. Even in that era, most people with MS (pwMS) lived much longer than 42, where the average life expectancy was reduced by approximately 8-10 years. In the current DMT era, we estimate that the reduction in life expectancy is much lower than this and will continue to improve. At a recent meeting, a Norwegian neurologist claimed that the life expectancy of Norwegians diagnosed in the modern era would only lose about three years of life expectancy. This fantastic improvement in outcome shows how DMTs and better care have improved things.
Sadly, many of the MS charities have made Jacqueline du Pré the poster child of MS, backed up by the media who love using a tragedy to sell newspapers and magazines and get social media and TV views. Saying this, I don’t want to detract from the remarkable person Jacqueline du Pré was. We should celebrate her short life, not for her dying of MS but for her talent as one of the greatest cello players of the modern era.
However, on the other hand, I also don’t think we should pull our punches or give pwMS and their families a rose-tinted version of the world. Many HCPs tell their patients that MS does not cause death and is not a terminal disease. This is incorrect.
Three-quarters of deaths in pwMS are from MS-related complications, the most common being aspiration pneumonia due to swallowing problems, septicaemia from urinary tract infections or pressure sores and pulmonary emboli from immobility. Sadly there is also an increased mortality in pwMS from suicide; the suicide rate in pwMS is between 2 and 7.5 times the background rate.
COVID-19 also exposed that pwMS who are disabled, have comorbidities and are on anti-CD20 therapies are at greater risk of severe COVID-19 and death from COVID-19. These risk factors will likely also play out with seasonal flu and other common respiratory pathogens.
Other uncommon causes of death in pwMS are falls and head injuries. One of my patients who lived alone went to the toilet one night and fell into her mirror, which shattered the glass that lacerated her arm and brachial artery, and she tragically died of blood loss. This is a freak accident, but if she wasn’t unsteady on her feet from MS-related cerebellar ataxia, she would not have fallen and would likely still be alive today.
PwMS are also at risk of complications from being on DMTs and other medications. Chronic immunosuppression comes with risks. Natalizumab causes PML, which can be fatal. Anti-CD20 therapies are associated with low immunoglobulin levels in some pwMS that increase your risk of severe infections requiring hospitalisations, which rarely will cause death. AHSCT and alemtuzumab are associated with several life-threatening opportunistic infections and secondary autoimmune diseases. The fingolimod, an S1P modulator, is also associated with life-threatening opportunistic infections and secondary malignancies. The life-threatening adverse events associated with fingolimod are likely to occur with the newer generation S1P modulators, but hopefully at a lower rate.
Even symptomatic therapies come with risks. Early in my career, one of our patients with MS sadly died from carbamazepine-induced Stevens-Johnson syndrome, an allergic reaction that affects the skin. The carbamazepine had been started for MS-related trigeminal neuralgia. I have also had a near death in a patient who developed autoimmune hepatitis on one of the interferon-beta preparations.
PwMS are also at increased risk of having or developing metabolic syndrome and vascular comorbidities. This is common to almost all chronic diseases associated with systemic inflammation, e.g. rheumatoid arthritis, SLE, etc. As a result, pwMS are at higher risk of myocardial infarction and stroke. Two relatively common causes of death in pwMS. Can we say these causes of death are unrelated to MS? I am not sure we can.
When fingolimod was first launched, several cases of sudden death were reported. This was thought to be due to cardiac conduction problems hence the increased vigilance with screening patients at baseline and not starting fingolimod or other newer generation S1P modulators (siponimod, ozanimod and ponesimod).
The fingolimod-sudden death cases highlighted a syndrome that had been largely forgotten, which I referred to as SUdden Death in MUltiple Sclerosis (SUDMUS), a play on SUDEP (SUdden Death in EPilepsy), which is due to the autonomic effects of seizures on cardiac conduction. A similar thing can happen in MS, typically due to brain stem lesions.
Cardiac conduction disorders are infrequently reported in acute MS relapse. I have never seen a case myself, but I refer to a short case report of a young person with acute onset of cerebellar symptomatology associated with sinus bradycardia (slow heartbeat). Her MRI showed one lesion in the left cerebellar hemisphere, which enhanced after contrast, and she had a non-enhancing midbrain lesion. No cardiac pathology was found, and her symptoms gradually improved after a 5-day course of corticosteroid therapy (Jurić et al. Newly onset sinus bradycardia in the context of multiple sclerosis relapse. Intern Med. 2012;51(9):1121-4).
In another case report, a patient with active relapsing-remitting MS died unexpectedly due to the sudden onset of cardiac arrhythmia. At post-mortem, there were large active demyelinating lesions in the medulla oblongata. As far as they know, this is the first case report linking sudden cardiac death to active MS on the histopathological level (Hengstman & Kusters. Sudden cardiac death in multiple sclerosis caused by active demyelination of the medulla oblongata. Mult Scler. 2011 Sep;17(9):1146-8).
Cardiovascular autonomic dysfunction is not uncommon in multiple sclerosis (MS) and is related to the involvement of the autonomic areas of the brain that supply the heart; these are located in the area of the brain stem called the medulla oblongata. It has been suggested that involvement in these areas may contribute to the occurrence of sudden death in MS.
A small number of patients with MS develop epilepsy, and a few unexplained deaths in MS could be due to SUDEP. Finally, a rare complication of brain stem relapses is pulmonary oedema or water on the lungs. The death of a young girl from this complication led to a new and profound insight into what MS is as a disease.
Interferon-beta
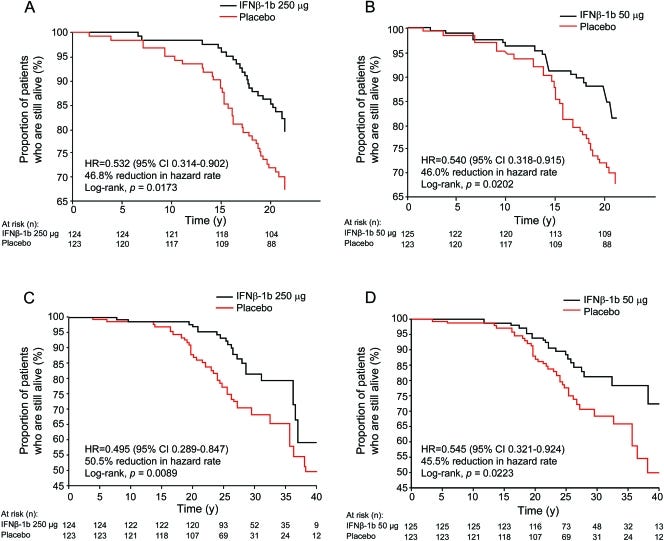
PwMS in the pivotal or original interferon-beta-1b study who had delayed access to interferon-beta-1b because they were randomised to receive placebo were 50% less likely to be alive at 21 years compared to study subjects with early access to treatment. This was almost certainly due to the impact of interferon-beta on MS rather than some other confounding factor. In the 69 pwMS for whom information on the relationship of death to MS was available, 78% were judged to have died from MS-related complications. This tells us that IFN-beta increases your life expectancy by reducing MS-related complications that can cause death in the future.
It would be best if you remembered that this was a placebo-controlled study, and the subjects on placebo were switched to active treatment after three years, with some subjects having to wait up to five years (trial recruitment was from June 1988 to May 1990; with placebo-treated subjects given free commercial supply as of October 1993). This study shows that delaying access to treatment by just 3-5 years has a significant impact on long-term outcomes.
Please note there is now overwhelming evidence from other controlled trials and real-life data to support the interferon-beta 21-year mortality data. The debate about early treatment and MS outcomes is so widely accepted that it has now shifted from early access to DMTs to early access to highly effective DMTs, i.e. flipping the pyramid. PwMS treated with more effective DMTs first-line do much better than those who are asked to wait (watchful waiting) or are escalated gradually up the DMT ladder (slow escalation). This is why, despite us designing the early treat-2-target NEDA trial, our centre couldn’t participate in the trial comparing maintenance-escalation vs flipping the pyramid. We don’t have equipoise; in other words, we don’t think it is ethical to randomise patients to a treatment arm that is less effective and hence will result in poorer long-term outcomes and probably poorer survival. This is why it is our current clinical practice to offer pwMS who have active disease and are eligible under the NHSE algorithm for treatment access to highly effective treatment first-line.
The Prineas lesion
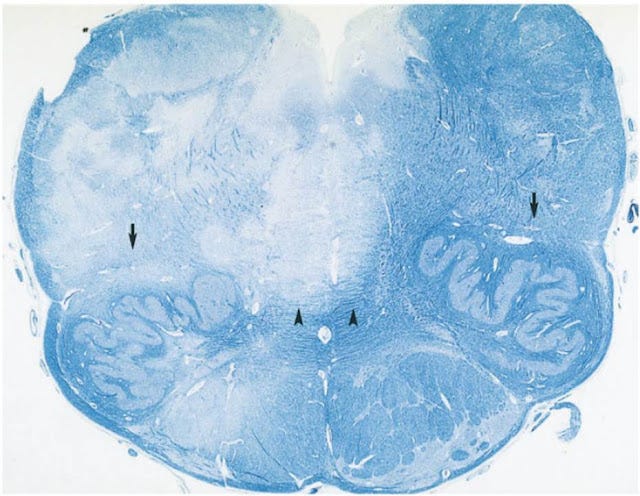
The earliest lesion in MS came from a patient who sadly died from SUDMUS. The lesion is referred to as the Prineas lesion after the neurologist who described the lesion. Yes, John Prineas was a neurologist and not a pathologist. In these lesions, there is a massive death of myelin-producing cells, called oligodendrocytes, by programmed cell death. The death of these cells appears to occur before T and B cells have arrived in the lesion. Finding out what is killing these cells will be an important piece of the jigsaw of finding out what causes MS.
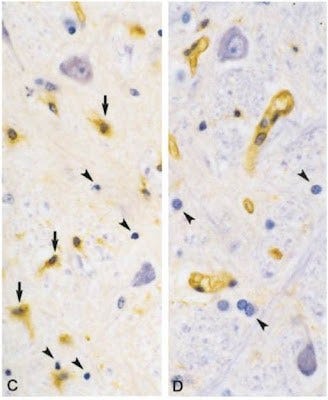
One of the most critical studies in the field of MS pathology was published in 2004, describing the hyperacute or very early MS lesion. The Barnette-Prineas paper changed our understanding of MS and continues to challenge the accepted wisdom or dogma today. They describe the pathological postmortem findings in 12 patients with RRMS who died during or shortly after the onset of a relapse. Changes not previously associated with the formation of new symptomatic lesions were observed in seven cases, i.e. extensive death of oligodendrocytes (the cells that produce myelin) by a cellular process called programmed cell death or apoptosis*and the extensive activation of another population of resident inflammatory cells called microglia (tissue scavengers).
The most remarkable observation was that there were very few or no lymphocytes in these lesions. Lymphocytes are the cells that are thought to drive autoimmunity and enter the brain and spinal cord from the peripheral blood. If these lesions don’t have T & B cells in them, we have to ask the obvious question: what is causing the death of the oligodendrocytes?
Before this paper, it was believed that lymphocytes orchestrated the first events in new MS lesion formation. This dogma arose from observations in the animal model of MS called EAE (experimental allergic encephalomyelitis); it is now evident that MS is not EAE.
The now famous 17-hour lesion above is visible in the critical part of the brain called the medulla, the part of the brain that controls vital functions like breathing. The myelin reduction in the left lesion appears pale or almost white. Compare this to the right side, where the normal myelin stains blue. In the high-power picture below, we see the myelin-producing cells, oligodendrocytes, showing the characteristic signs of programmed cell death or apoptosis. There is a lack of lymphocytes in this area of the lesion.
What is killing these oligodendrocytes? Could it be a virus? Is the influx of lymphocytes seen in older MS lesions a secondary response to the inciting agent? Too many questions and not enough answers. This paper raises serious questions about the hypothesis that MS is an autoimmune disease. I don’t accept MS as a primary autoimmune disease; I prefer the viral hypothesis that the inflammation we see in MS is a secondary response to a virus. This paper has been very controversial, and the world’s pathologists still can’t agree on the interpretation of the findings. The paper has been criticised by some very eminent pathologists, whereas others have kept a low profile and not openly expressed an opinion.
This Newsletter highlights several different issues. (1) MS is a disease associated with disability and early death. The majority of deaths in MS are MS-related, particularly in relation to disability (swallowing, bladder), immobility (pressure sores, deep vein thrombosis), mental health (suicide), inflammation (metabolic syndrome and vascular disease), secondary to side effects of the treatment of MS (infections and rare an adverse events) and rarely due to SUDMUS from brain stem lesions. (2) The prognosis and management of MS are improving, and as a result, outcomes in the modern era are vastly different from those in the pre-DMT era. (3) A lot can be learnt from studying the brains of pwMS who die from SUDMUS. I would therefore urge you all to sign-up for brain donation. The very early Prineas lesion supports the viral aetiology of MS; without the study of pwMS dying from MS, we would not be able to advance the field of MS research.
*apoptosis /ap·op·to·sis/ (ap″op-to´sis) a pattern of cell death affecting single cells, marked by shrinkage of the cell, condensation of chromatin, and fragmentation of the cell into membrane-bound bodies that are eliminated by phagocytosis. Often used synonymously with programmed cell death.
The study describes the clinical and pathological findings in 12 patients with relapsing and remitting multiple sclerosis who died during or shortly after the onset of a relapse. Pathological changes not previously associated with the formation of new symptomatic lesions were observed in seven cases, namely, extensive oligodendrocyte apoptosis and microglial activation in myelinated tissue containing few or no lymphocytes or myelin phagocytes. No current laboratory model of multiple sclerosis, in particular, experimental allergic encephalomyelitis, is known with these features, which raises the possibility of some novel process underlying new lesion formation in multiple sclerosis.
Objective: To examine the effects of interferon beta (IFNβ)-1b on all-cause mortality over 21 years in the cohort of 372 patients who participated in the pivotal randomized clinical trial (RCT), retaining (in the analysis) the original randomized treatment-assignments. Methods: For this randomized long-term cohort study, the primary outcome, defined before data collection, was the comparison of all-cause mortality between the IFNβ-1b 250 μg and placebo groups from the time of randomization through the entire 21-year follow-up interval (intention-to-treat, log-rank test for Kaplan-Meier survival curves). All other survival outcomes were secondary. Results: After a median of 21.1 years from RCT enrollment, 98.4%(366 of 372) of patients were identified, and, of these, 81 deaths were recorded (22.1% [81 of 366]). Patients originally randomly assigned to IFNβ-1b 250 μg showed a significant reduction in all-cause mortality over the 21-year period compared with placebo (p = 0.0173), with a hazard ratio of 0.532 (95% confidence interval 0.314-0.902). The hazard rate of death at long-term follow-up by Kaplan-Meier estimates was reduced by 46.8% among IFNβ-1b 250 μg-treated patients (46.0% among IFNβ-1b 50 μg-treated patients) compared with placebo. Baseline variables did not influence the observed treatment effect. Conclusions: There was a significant survival advantage in this cohort of patients receiving early IFNβ-1b treatment at either dose compared with placebo. Near-complete ascertainment, together with confirmatory findings from both active treatment groups, strengthens the evidence for an IFNβ-1b benefit on all-cause mortality. Classification of evidence: This study provides Class III evidence that early treatment with IFNβ-1b is associated with prolonged survival in initially treatment-naive patients with relapsing-remitting multiple sclerosis.
Objectives: Compared with controls, multiple sclerosis (MS) patients die, on average, 7–14 years prematurely. Previously, we reported that, 21 years after their participation in the pivotal randomised, controlled trial (RCT) of interferon β-1b, mortality was reduced by 46–47% in the two groups who received active therapy during the RCT. To determine whether the excessive deaths observed in placebo-treated patients was due to MS-related causes, we analysed the causes-of-death (CODs) in these three, randomised patient cohorts. Design: Long-term follow-up (LTF) of the pivotal RCT of interferon β-1b. Setting: Eleven North American MS-centres participated. Participants: In the original RCT, 372 patients participated, of whom 366 (98.4%) were identified after a median of 21.1 years from RCT enrolment. Interventions: Using multiple information sources, we attempted to establish COD and its relationship to MS in deceased patients. Primary outcome: An independent adjudication committee, masked to treatment assignment and using prespecified criteria, determined the likely CODs and their MS relationships. Results: Among the 366 MS patients included in this LTF study, 81 deaths were recorded. Mean age-at-death was 51.7 (±8.7) years. COD, MS relationship, or both were determined for 88% of deaths (71/81). Patients were assigned to one of nine COD categories: cardiovascular disease/stroke; cancer; pulmonary infections; sepsis; accidents; suicide; death due to MS; other known CODs; and unknown COD. Of the 69 patients for whom information on the relationship of death to MS was available, 78.3% (54/69) were adjudicated to be MS related. Patients randomised to receive placebo during the RCT (compared with patients receiving active treatment) experienced an excessive number of MS-related deaths. Conclusions: In this long-term, randomised, cohort study, MS patients receiving placebo during the RCT experienced greater all-cause mortality compared to those on active treatment. The excessive mortality in the original placebo group was largely from MS-related causes, especially, MS-related pulmonary infections.
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry nor Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your healthcare professional, who will be able to help you.