Case study
Dear Prof Giovannoni, I am not sure if you remember me. I was your patient when you worked at the National Hospital for Neurology and Neurosurgery. I have been in a wheelchair for over seven years and have spasticity in my legs and severe bladder and bowel problems. I am now being looked after by my local hospital.
Prof G’s opinion
The above story is a familiar one of a patient with advanced MS being lost to follow-up by a specialist MS centre and being managed locally by a general neurologist or usually by the general practitioner. Ideally, this patient needs to be looked after by an MS specialist nurse, with a specialist MS neurologist involved when necessary. In short, there is a lot that can be done for this patient. Where do I begin?
Now that the COVID-19 epidemic in the UK is over, in the sense that there are no restrictions, I have restarted doing MS roadshows again. I went to talk to a group of people with MS and their families in North London last night. Many of the attendees had quite advanced MS and were using wheelchairs. It was clear from some of their comments that many of them are being let down by their MS services in that they don’t find their annual follow-up appointments very helpful, similar to the patient above.
Sadly the attitude of the general neurologist above is pervasive and is not helped when senior and very influential academics make comments such as ‘once you have clinically manifest progressive MS with disability the therapeutic window has probably shut’. These attitudes dampen the spirits of many people living with advanced MS and challenge some of the emerging concepts about smouldering MS and the research we are involved in.
We now realise that multiple sclerosis is a length-dependent axonopathy, i.e. the longer the nerve pathway is, the more likely it is affected by multiple MS lesions and the earlier it loses reserve capacity. This explains why lower limb function is affected before upper limb function and underpins the concept that there are multiple therapeutic windows in MS. In other words, even if you have lost lower limb function, you still have upper limb function to protect, not to mention speech, swallowing, cognition, etc. This insight underpins our #ThinkHand campaign and why we are doing the CHARIOT-MS (ClinicalTrials.gov Identifier: NCT04695080) and ORATORIO-HAND (ClinicalTrials.gov Identifier: NCT04035005) studies and the UNDER&OVER arm and hand rehabilitation programme. In short, I think it is never too late to modify the course of MS.
A few years ago, during the #ThinkHand campaign, one of my colleagues presented a patient with advanced MS (EDSS 7.5 = wheelchair-bound) who was still on interferon-beta. In keeping with NHS England guidelines, she had to stop treating this patient with interferon-beta. This patient presented with double vision three months later due to a brain-stem relapse. An MRI with contrast showed multiple Gd-enhancing lesions indicative of reactivation of MS disease activity, or rebound, post-interferon-beta. As this patient was not eligible to be restarted on a licensed DMT, we recommended that the patient be treated off-label with generic subcutaneous cladribine. Our position was that although the patient may be wheelchair-bound with advanced MS, she still had upper limb (arm & hand) and bulbar (swallowing and speech) functions to preserve. Were we wrong to treat this patient and assume that the therapeutic window to modify her disease course has shut? This patient is not unique; I am prepared to bet that almost every MS expert has had similar experiences with patients in their practice.
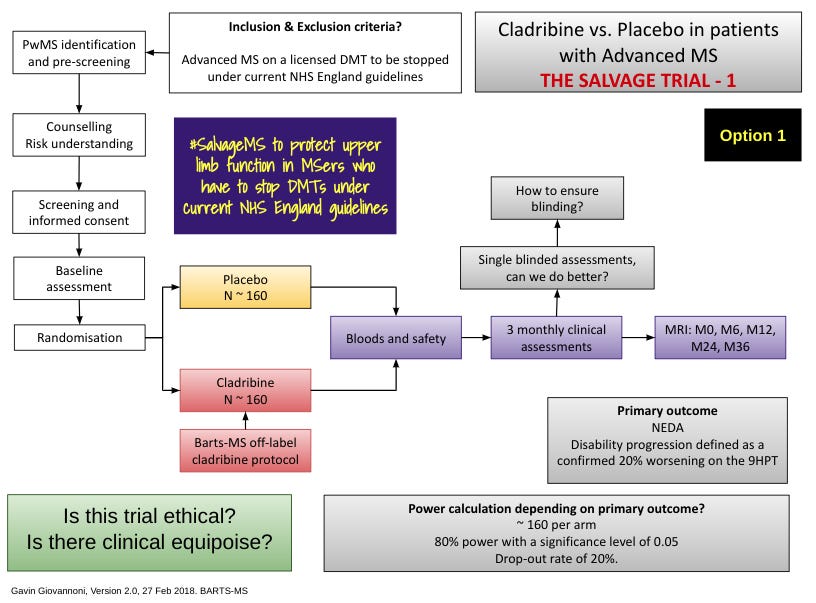
As a result of these insights, we need to challenge therapeutic nihilism in advanced MS and the NHS England DMT stopping criteria. This is why I have proposed doing a randomised controlled trial to evaluate whether or not MS is modifiable in the population of patients who have to stop their licensed DMT. Are these ‘unsalvageable’ salvageable? I named this trial the SALVAGE study.
I propose randomising pwMS who are forced to stop their existing DMT to treatment with possible generic cladribine or placebo with the primary outcome being NEIDA (no evident inflammatory disease activity) or possibly disease worsening as defined using upper limb function, i.e. the 9-hole peg test. The plan is to make this an event-driven trial, and if someone reaches the study end-point, they can be unblinded and offered a course of cladribine if they were previously treated with placebo. If they had broken through on cladribine, they could be offered a further course of treatment or another off-label salvage therapy, for example, rituximab biosimilar.
What I say to my colleagues and the wider medical community, instead of assuming MS is not modifiable beyond a certain stage, should we not at least try and salvage upper limb function in people with more advanced MS?
The real question is, do we have the equipoise to do this study, i.e. is it ethical?
In a small French study, stopping DMTs in people with SPMS, 35% of the cohort had relapses, or MRI activity, in the follow-up period (~5 years). Similarly, in the MS-BASE study below, of 485 DMT-stoppers vs. 854 DMT-stayers followed for at least three years, the time to confirmed disability worsening was significantly shorter among the subjects who stopped their DMT compared to those who stayed on treatment. The data from these two studies would indicate that we should ignore NHS England because their stopping criteria are not evidence-based. Instead, we should allow the patient to decide whether or not they want to stop their DMT or continue with it.
What do you think?
Another message I gave last night was that in addition to DMTs, there is a lot that can be done for people with advanced MS in terms of symptomatic treatment and preventing MS-related complications (infections, falls, fractures, etc.…). I discussed the holistic management of MS and introduced them to marginal gains.
For more information on marginal gains, please read my Newsletter on ‘Brain Health: how important is it? (6-Oct-2022)’.
I strongly recommend that to get the most out of your annual MS review, you must prepare for the consultation using one of the online resources such as ‘Your MS Questionnaire’ or ‘My MS Priorities’.
Please see MS-Selfie Newsletter: ‘What are your priorities in relation to 'Your MS'? (4-Oct-2021)
If you are interested in more information on MS being a length-dependent axonopathy, I suggest you read the following paper.
Trials of anti-inflammatory therapies in non-relapsing progressive multiple sclerosis (MS) have been stubbornly negative except recently for an anti-CD20 therapy in primary progressive MS and an S1P modulator siponimod in secondary progressive MS. We argue that this might be because trials have been too short and have focused on assessing neuronal pathways, with insufficient reserve capacity, as the core component of the primary outcome. Delayed neuroaxonal degeneration primed by prior inflammation is not expected to respond to disease-modifying therapies targeting MS-specific mechanisms. However, anti-inflammatory therapies may modify these damaged pathways, but with a therapeutic lag that may take years to manifest. Based on these observations we propose that clinically apparent neurodegenerative components of progressive MS may occur in a length-dependent manner and asynchronously. If this hypothesis is confirmed it may have major implications for the future design of progressive MS trials.
French Study
BACKGROUND AND PURPOSE: The benefits of immunomodulatory treatments in secondary progressive multiple sclerosis (SPMS) are unclear, calling into question their continuation. In the present observational study, we investigated the effect of treatment withdrawal on the clinical course of SPMS.
METHODS: We included 100 consecutive patients with SPMS who regularly attended our multiple sclerosis clinic. Inclusion criteria were (i) secondary progressive phenotype for at least 2 years, (ii) immunomodulatory treatment for at least 6 months and (iii) treatment stopped with no plans to switch to another. Clinical and magnetic resonance imaging (MRI) data before and after treatment discontinuation were assessed. Factors associated with relapses and/or MRI activity were identified.
RESULTS: Mean treatment duration was 60.4 ± 39.3 months, and mean follow-up duration after treatment withdrawal was 62.4 ± 38.4 months. The annualized relapse rate remained stable at 1 and 3 years after treatment withdrawal [0.09, 95% confidence interval (CI), 0.05-0.17 and 0.07, 95% CI, 0.05-0.11, respectively], relative to the 3 years prior to treatment withdrawal (0.12, 95% CI, 0.09-0.16). Sixteen patients experienced a relapse and 19 had a gadolinium-positive MRI scan without relapse during follow-up. A gadolinium-positive MRI scan within the previous 3 years before treatment withdrawal and Expanded Disability Status Scale score of <6 were positively associated with relapse and/or MRI activity after discontinuation (P = 0.0004 and P = 0.03, respectively).
CONCLUSION: In this retrospective study, including a limited number of patients with SPMS, the annualized relapse rate remained stable after treatment withdrawal, relative to before treatment withdrawal. Further prospective studies are needed to confirm this result and provide evidence-based guidelines for daily practice.
MS-BASE STUDY
BACKGROUND: Discontinuation of injectable disease-modifying therapy (DMT) for multiple sclerosis (MS) after a long period of relapse freedom is frequently considered, but data on post-cessation disease course are lacking.
OBJECTIVES: (1) To compare time to first relapse and disability progression among ‘DMT stoppers’ and propensity-score matched ‘DMT stayers’ in the MSBase Registry; (2) To identify predictors of time to first relapse and disability progression in DMT stoppers.
METHODS: Inclusion criteria for DMT stoppers were: age ≥18 years; no relapses for ≥5 years at DMT discontinuation; follow-up for ≥3 years after stopping DMT; not restarting DMT for ≥3 months after discontinuation. DMT stayers were required to have no relapses for ≥5 years at baseline, and were propensity-score matched to stoppers for age, sex, disability (Expanded Disability Status Score), disease duration and time on treatment. Relapse and disability progression events in matched stoppers and stayers were compared using a marginal Cox model. Predictors of first relapse and disability progression among DMT stoppers were investigated using a Cox proportional hazards model.
RESULTS: Time to first relapse among 485 DMT stoppers and 854 stayers was similar (adjusted HR, aHR=1.07, 95% CI 0.84 to 1.37; p=0.584), while time to confirmed disability progression was significantly shorter among DMT stoppers than stayers (aHR=1.47, 95% CI 1.18 to 1.84, p=0.001). The difference in hazards of progression was due mainly to patients who had not experienced disability progression in the prebaseline treatment period.
CONCLUSIONS: Patients with MS who discontinued injectable DMT after a long period of relapse freedom had a similar relapse rate as propensity score-matched patients who continued on DMT, but higher hazard for disability progression.
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry nor Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have problems, please tell your healthcare professional, who will be able to help you.













Is your MS salvageable?