Primary progressive multiple sclerosis
Four buses have arrive together. How will we use the data to improve the lives of pwPPMS?
I have used the waiting for a bus analogy in the past.
The “three bus analogy” is a popular British expression referring to waiting for a bus for a long time, only to have three buses arrive at once. It is often used to describe any scenario in which a resource is scarce and then suddenly abundant. I am not sure if people with MS (pwMS) and the other MS stakeholders realise what the implications of three recent ocrelizumab-related PPMS trials and one tolebrutinib PPMS trial mean for the field.
Bus 1 - GAVOTTE Study
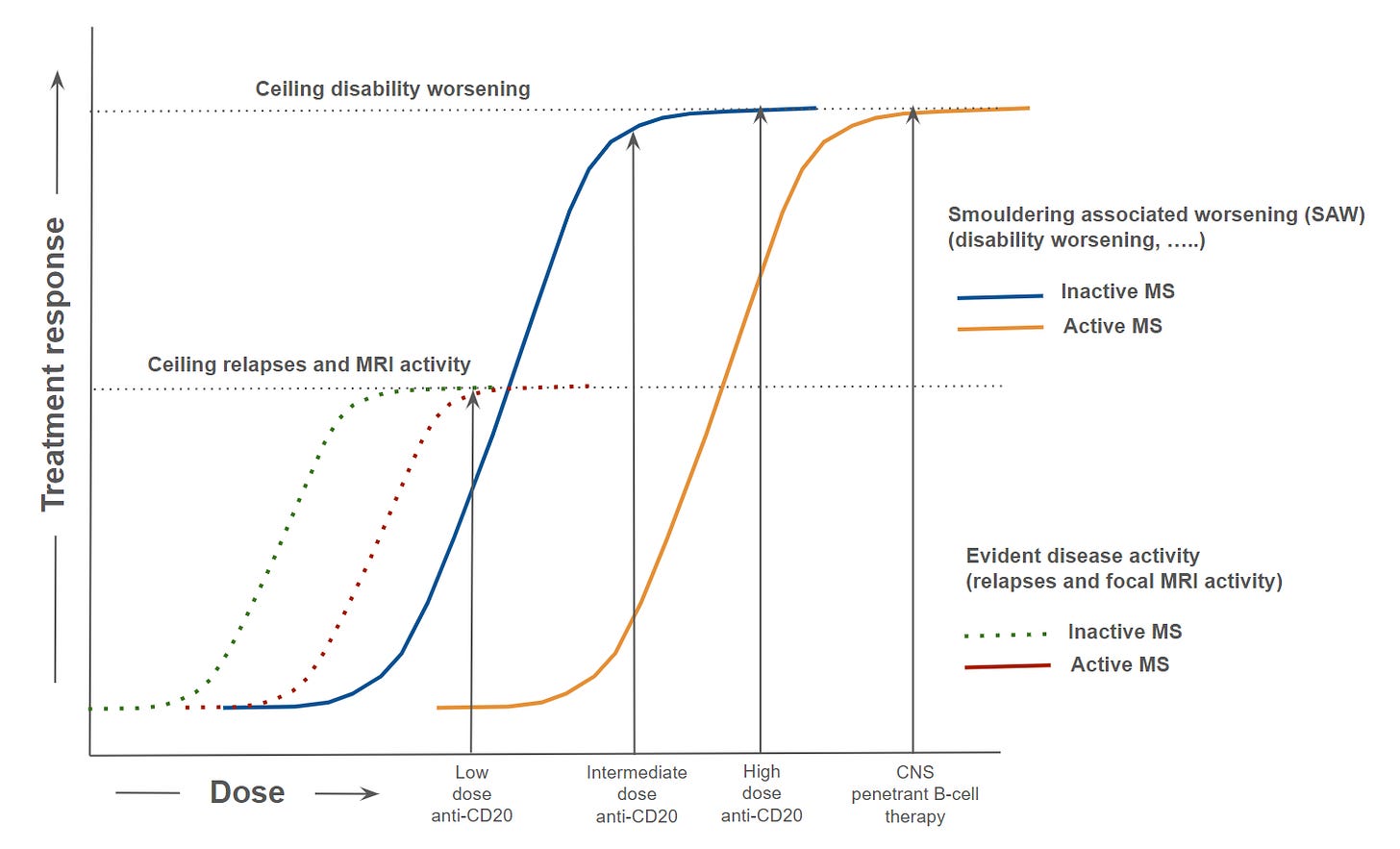
First, there was the news that the high-dose versus standard-dose ocrelizumab PPMS study, or GAVOTTE study, was negative, i.e. there was no difference in disability outcomes between the two arms. However, the active group of subjects with PPMS, i.e., those with baseline Gd-enhancing lesions, did better with higher doses than with standard doses. I am aware that we must always be careful when looking at post-hoc analyses, as they are often underpowered and part of a data-torturing exercise. However, a similar observation was seen in the relapsing or MUSETTE study. These observations will not affect the license or how Ocrelizumab is used, but it does inform how we think about the biology of MS in relation to B-cell targeted therapies. This onservation of a better response in relation to smoudlering MS in relation to baseline disease activity is supported by the observation that high-dose rituximab and standard-dose ocrelizumab are superior at preventing disability worsening in subjects with active PPMS, as defined by the presence of Gd-enhancing lesions at baseline. Is this because Gd-enhancing lesions are an indicator or message from the immune system that whatever is causing MS is active and modifiable by anti-CD20 therapy? Could this be related to EBV? I have argued in the past that rebound on natalizumab withdrawal is likely to be due to CNS EBV reactivation and is blocked by anti-CD20 therapy. Could Gd-enhancing lesions in any siuation, not just post-natalizumab, indicate the same pathology?
The fact that pwMS with active MS do better with higher doses of anti-CD20 therapy suggests that the dose-response curve is complex and that higher doses may be needed for more active patients, i.e., that standard-dose ocrelizumab is not on the shoulder of the dose-response curve (see figure below). It is for this reason why I think higher doses of ocrelizumab are better than lower doses. I think this is due to deeper B-cell depletion and possibly greater CNS effects with higher doses. This is not a trivial issue and applies to other diseases such as lupus nephritis. In general, rituximab has only a modest or no effect on lupus nephritis. However, the recent approval of Gazyva (obinutuzumab) for active lupus nephritis illustrates this. Obinutuzumab is one of the most effective deep-tissue-depleting anti-CD20 monoclonal antibodies on the market, underscoring the importance of achieving deep tissue depletion for treating B-cell-mediated autoimmune disease or EBV-driven autoimmunity. I am sure the same lessons apply to MS and makes a strong case for CD19-targeting CAR T-cells as a treatment for lupus, MS and other autoimmune diseases.
Bus 2 - O’HAND Study
The O’HAND study, which I have discussed in detail in previous newsletters is the second bus or trial of importance. In short, O’HAND has shown that the original ORATORIO study was not a false-positive result, i.e. ocrelizumab is effective as a treatment for active PPMS, and there was also a treatment effect, albeit lower, in inactive PPMS. However, more importantly, pwPPMS who are wheelchair users benefited from ocrelizumab. This trial is a game-changer: we now know that even people with advanced MS will benefit from DMTs, and the primary outcome in progressive MS trials will likely change to include the 9-hole peg test. I have mentioned that the O’HAND will hopefully impact the stopping criteria for ocrelizumab as well. At present, we are being forced to stop ocrelizumab in pwPPMS who become wheelchair users. This will now be unethical, given that O’HAND shows that ocrelizumab slows the loss of hand and upper-limb function.
Please see:
Bus 3 - FENTREPID Study
The FENTREPID study has shown that fenebrutinib is non-inferior to ocrelizumab for the treatment of PPMS. However, across the piste of outcome measures, fenebrutinib looks better or superior to ocrelizumab. Interestingly, if the primary outcome in this trial was the same as that used in the O’HAND study, fenebrutinib would have been shown to be superior to ocrelizumab. This is because including the timed-25-foot walk (T25FW) in the composite for the primary outcome introduced noise. This is a sad state of affairs as we have known for some time, in fact, since the EXPAND siponimod SPMS trial, that the T25FW is too noisy and not a good outcome measure for MS. The good news is that Maria Pia-Sormani and colleagues have done a meta-analysis across numerous phase 3 studies showing the poor performance of the T25FW. I hope regulators will take this analysis into consideration when assessing the efficacy of fenebrutinib for PPMS.
I am currently presenting both the O’HAND and FETREPID results at the Association of British Neurology annual meeting in Birmingham (click here to download ABN posters). Despite my enthusiasm for these studies the response from my colleagues was underwhelming. I think they have been worn down by the riches of DMT data presented over the last 30 years for treating relapse-onset MS. What is not being noticed is that things are also changing for PPMS, a previously neglected group of pwPPMS.
Bus 4 - PERSEUS study
Sadly, bus 4 or the PERSEUS trial (NCT04458051) designed to answer whether tolebrutinib could alter the course of PPMS was negative. This study did not meet its primary endpoint of delaying the time to onset of 6-month composite confirmed disability progression compared to placebo. This failure is in contrast to the HERCULES trial, where tolebrutinib successfully delayed disability progression in non-relapsing Secondary Progressive MS (nrSPMS). This discordance—success in secondary progression but failure in primary progression— has triggered debate regarding the biological distinctions between these two forms of progressive disease. It suggests that, while they share features of smouldering inflammation, the specific drivers or the timing of required therapeutic intervention may differ fundamentally. I don’t agree with this and think the differences are due to bad luck; when we look at the PERSEUS trial in more detail, we see a treatment signal similar to that seen in the HERCULES trial. This supports other data that there are no differences between the biology of nrSPMS and PPMS.
I hope you agree with me on the importance of the first three buses, or trials, for the treatment of PPMS. The fourth bus may be empty, but it doesn’t change the message that PPMS is modifiable beyond EDSS 6.5 and that CNS penetrant therapies such as fenebrutinib will change the outlook for pwPPMS.
Paper - T25FW
Disability worsening is the critical long-term outcome in multiple sclerosis, yet the Expanded Disability Status Scale incompletely captures neurological deterioration and has limited sensitivity in the short time windows of clinical trials. Composite endpoints incorporating functional measures have been proposed to address these limitations, but whether they reliably improve detection of treatment effects has not been established across trials. We conducted a post-hoc analysis of individual patient data from ten phase III randomised controlled trials (ASCEND, BRAVO, CONFIRM, DEFINE, EXPAND, INFORMS, OLYMPUS, OPERA I/II, and ORATORIO; n = 9,369), spanning relapsing-remitting and progressive multiple sclerosis. Confirmed disability worsening was defined using harmonised criteria with the MSprog package and confirmed at 24 weeks. Treatment effects were estimated using Cox proportional hazards models and combined across trials in a one-stage individual patient data framework. Composite endpoints were constructed from the Expanded Disability Status Scale, the timed 25-foot walk test, and the nine-hole peg test using logical unions (OR-type), intersections (AND-type), and majority-vote structures. Sensitivity to treatment effect was quantified using Z-scores (the ratio of the pooled log-hazard ratio to its standard error) and compared to the Expanded Disability Status Scale reference using interaction tests. Event rates varied across components: the timed walk test generated the highest rates (up to 46.8%) while the nine-hole peg test generated the lowest (as low as 2.1%). OR-type composite endpoints showed weaker treatment effects than the Expanded Disability Status Scale alone, with the largest reductions in sensitivity observed for endpoints incorporating the timed walk test ( Δ Z up to +2.26; interaction p = 0.004). These findings were confirmed across disease subtypes and were pronounced in relapsing-remitting trials, where no composite endpoint outperformed the Expanded Disability Status Scale. In progressive multiple sclerosis, the combination of the Expanded Disability Status Scale and the nine-hole peg test showed numerically stronger treatment effects ( Δ Z = −1.65), though interaction tests did not reach statistical significance (p = 0.051). Composite endpoints do not systematically improve treatment effect detection in multiple sclerosis trials. Increased event capture driven by the timed walk test introduces noise that dilutes the treatment signal rather than amplifying it, highlighting that event rate and endpoint quality are not interchangeable. Upper limb function assessed by the nine-hole peg test provides complementary and specific information, particularly in progressive disease. The combination of global disability and upper limb measures represents a promising direction for future endpoint development in progressive multiple sclerosis trials, warranting validation.
Accidental readers
If you have been forwarded this email and are not an MS-Selfie subscriber, please consider subscribing and helping MS-Selfie expand its resources for the broader MS community. MS-Selfie relies on subscriptions to fund its curated MS-Selfie microsite, MS-Selfie books, MS-Selfie Infocards, and other activities that extend beyond the MS-Selfie Substack newsletters.
Subscriptions and donations
MS-Selfie newsletters and access to the MS-Selfie microsite are free. In comparison, off-topic Q&A sessions are restricted to paying subscribers. Subscriptions are being used to run and maintain the MS Selfie microsite and other related activities, as I don’t have time to do this myself. You must be a paying subscriber to ask questions unrelated to the newsletters or podcasts. If you can’t afford to become a paying subscriber, please email a request for a complimentary subscription (ms-selfie@giovannoni.net).
Questions
If you have questions unrelated to the newsletters or podcasts, please email them to ms-selfie@giovannoni.net. Prof. G will try to answer them as quickly as possible.
Important Links
🖋 Medium
General Disclaimer
Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Queen Mary University of London or Barts Health NHS Trust. The advice is intended as general and should not be interpreted as personal clinical advice. If you have any problems, please tell your healthcare professional, who can help you.


The fact that Fenebrutinib is effective in non-active PPMS is encouraging, but the efficacy still remains low and the risk profile is not that good.
I’m hopeful that the next buses will be :
- IMU-838 (vidofludimus calcium) : start of phase 3 trial in PPMS in H2 2026
- Foralumab (anti-CD3) : phase 2a results for SPMS expected by this summer
- Frexalimab (anti-CD40L) : phase 3 results for SPMS expected in late 2026 or early 2027
- Remibrutinib (BTKi) : phase 3 results for RRMS expected by this summer ; highly selective and with a very good risk profile so far. A phase 3 trial for SPMS also started in late 2025.
- TYK2 inhibitors (SUDO-550, A-005, etc.) entering phase 2 soon
- And of course anti-BCMA CAR-T therapies (CT103A, AZD0120)
It would be fabulous if one of the DMTs could become available to this 74 year old female whose mobility has declined rapidly in the last 18 months. However, I’m still able to walk short distances. Improvement would be wonderful but even plateauing would be good. I fear time is running out for me.😢