BTK inhibitors - will they deliver?
I am going to be an optimist and give this new class of treatment a 75% chance of success (range 60-90%), mainly due to their anti-B-cell effects, and a 25% chance of failure. Why?
The main focus in studying and managing MS in 2022 is smouldering disease, i.e. the processes that are occurring beyond focal inflammatory lesion activity. Can we develop and add-on treatments to modify the processes causing people with MS (pwMS) to get worse despite being NEIDA (no evident inflammatory disease activity)? If the concept of smouldering MS is new to you please read my other newsletters on the subject.
The question on everyone’s mind is how will the BTK (Bruton Tyrosine Kinase) inhibitor race turnout; will it be mass suicide with them failing as a class or will they usher in the next generation of innovative MS treatments?
Our interest in BTK inhibitors started about 7 years ago when the Mouse Doctor and I almost managed to get Abbvie to fund an investigator-led study of Ibrutinib in MS. However, it was not to be as Abbvie’s partner Janssen blocked the grant. Janssen was concerned that it was too risky to test Ibrutinib in MS because of the off-target effects of Ibrutinib and its potential for serious adverse events. I suspect they were right as Ibrutinib is a dirty drug and not a very selective BTKi as it also inhibits several other kinases.
Our hypothesis was simple; we wanted a CNS penetrant drug to target B-cells and plasma cells in the CNS of pwMS. We were buoyed by the observation that several people with CNS B-cell lymphomas were having dramatic responses to Ibrutinib. Although it was never to be we have continued our search for a CNS penetrant anti-B-cell and anti-plasma cell agent and eventually, we managed to convince Takeda to fund a trial of their CNS penetrant second-generation proteasome inhibitor Ixazomib in MS (SIZOMUS Study). Although initiation and recruitment for this study were delayed by COVID-19 we are now in the second phase of recruitment.
Despite our failure to get Ibrutinib, a first-generation BTK-inhibitor, into MS Big Pharma has taken up the challenge and there are now at least five companies with BTKi programmes in MS (Merck KGaA, Sanofi-Genzyme, Roche, Novartis and Biogen).
BTKIs work in MS because they inhibit B-cell activation, by preventing signalling via the B-cell or immunoglobulin receptor. There is phase 2 data for two of these agents confirming this (evobrutinib, Merck; tolebrutinib, Sanofi-Genzyme). However, most people are not aware that BTKi also inhibits macrophage and microglial activation via the Fc receptor (FcR) signalling pathway. Therefore CNS penetrant BTK inhibitors, which apply to at least three of the five BTKi’s referred to above, will also target the so-called ‘hot’ or activated microglial response and test the hypotheses whether or not this response is favourable in MS.
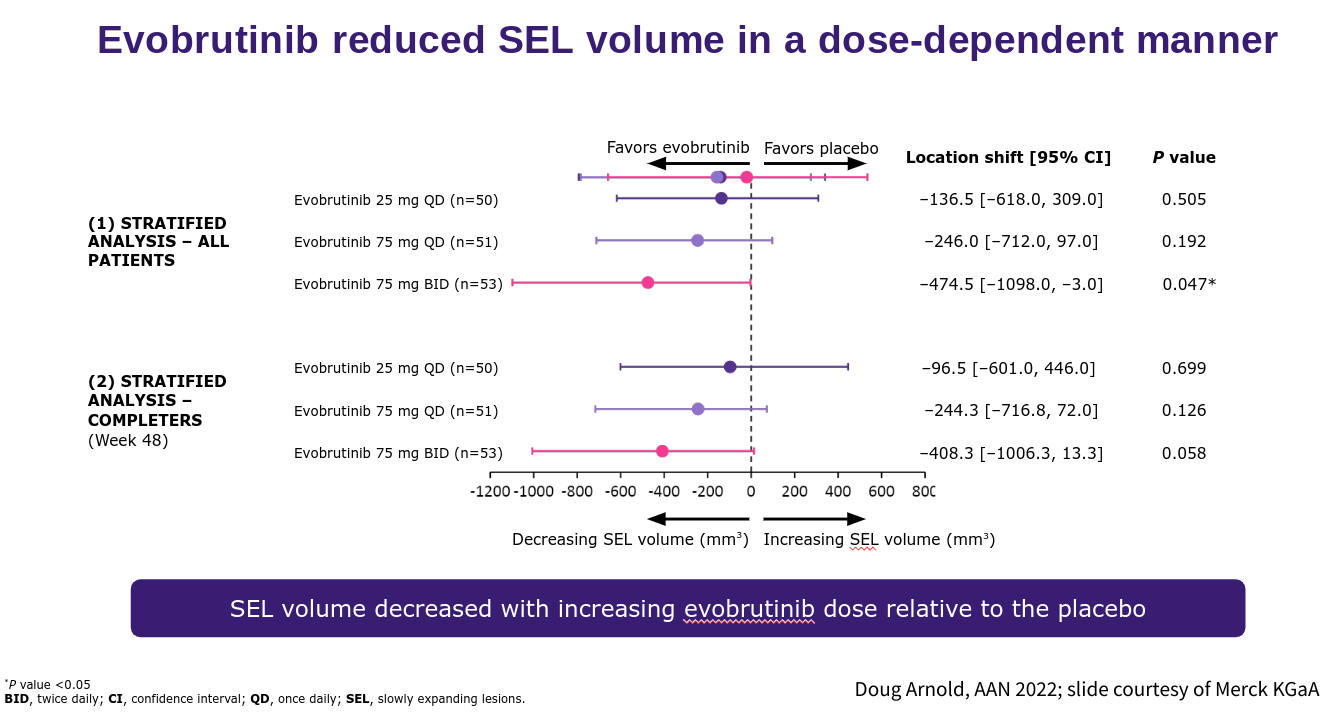
The problem will be dissecting the contribution of the anti-B-cell response from the anti-microglial response in terms of efficacy. Clearly, this will be important in view of some of the issues I raised around the ‘hot microglial’ response being potentially beneficial in the pathogenesis of MS. I envisage BTKi being very effective in stopping relapses and focal MRI activity the big question will be about the impact of BTKi on the smouldering component of MS. The good news is that at the recent AAN meeting Doug Arnold presented compelling data that evobrutinib, in a dose-response manner, slowed the expansion or increasing volume associated with the so-called slowly expanding lesion (SEL). We know that SELs are bad news for pwMS and are associated with a worse outcome. This early data would indicate that BTKi’s are targeting at least one process associated with smouldering MS and hence as a class are offering hope to pwMS who have overt smouldering disease.
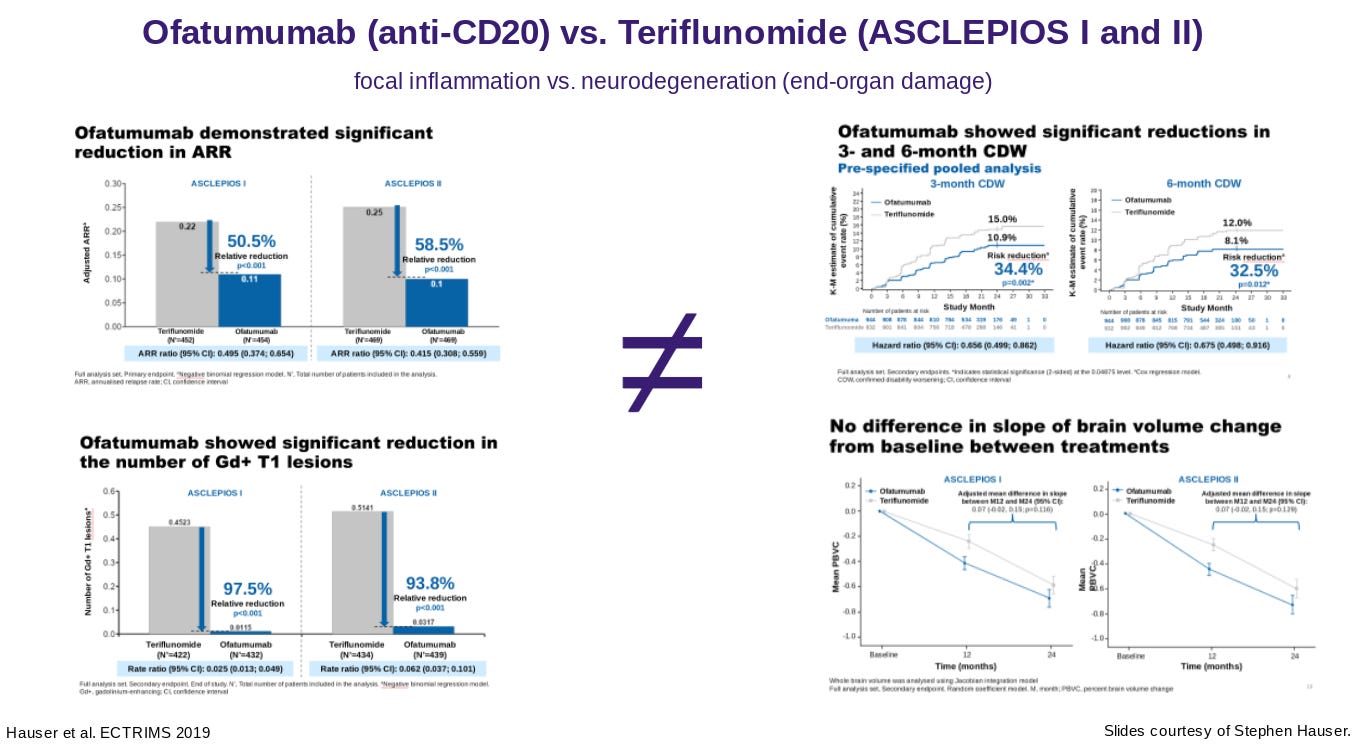
I note that many of the phase 3 studies will be testing BTKi against teriflunomide. Clearly, BTKi’s are likely to beat Teri in terms of their impact on relapses and focal MRI activity, but Pharma may be taking a chance in the hope of beating Teriflunomide in terms of its impact on the end-organ or the smouldering component of MS. Don’t forget ofatumumab and teriflunomide had the same effect on brain volume loss when they were compared head-2-head in the ASCLEPIOS I and ASCLEPIOS II studies despite ofatumumab being clearly superior to teriflunomide in suppressing relapses and focal MRI activity. Clearly, teriflunomide is having an impact on the end-organ that is independent of its anti-inflammatory effects on relapses and focal MRI activity. Could it be the antiviral and in particular the anti-EBV effect of teriflunomide that explains its impact on brain volume loss and disability progression in MS?
It is clear to me that BTK is a very exciting and important treatment target in MS and the phase 3 trials will provide additional evidence beyond the B-cell on whether or not we should be targeting macrophage and microglial activation via their Fc-receptors.
I am going to be an optimist and give this new class of treatment a 75% chance of success (range 60-90%), mainly due to their anti-B-cell effects, and a 25% chance of failure. Failure will be due to unintended effects of inhibiting microglia, yet to be identified off-target effects and as yet unidentified toxicity. What is important is that we are testing a hypothesis about the smouldering component of MS, which I consider to be the real MS and this is why I am so excited to be part of the story.
Paper 1
Background: Bruton's tyrosine kinase (BTK) regulates the functions of B cells and myeloid cells that are implicated in the pathogenesis of multiple sclerosis. Evobrutinib is a selective oral BTK inhibitor that has been shown to inhibit B-cell activation both in vitro and in vivo.
Methods: In this double-blind, randomized, phase 2 trial, we assigned patients with relapsing multiple sclerosis to one of five groups: placebo, evobrutinib (at a dose of 25 mg once daily, 75 mg once daily, or 75 mg twice daily), or open-label dimethyl fumarate (DMF) as a reference. The primary end point was the total (cumulative) number of gadolinium-enhancing lesions identified on T1-weighted magnetic resonance imaging at weeks 12, 16, 20, and 24. Key secondary end points included the annualized relapse rate and change from baseline in the score on the Expanded Disability Status Scale (EDSS).
Results: A total of 267 patients were randomly assigned to a trial group. The mean (±SD) total number of gadolinium-enhancing lesions during weeks 12 through 24 was 3.85±5.44 in the placebo group, 4.06±8.02 in the evobrutinib 25-mg group, 1.69±4.69 in the evobrutinib 75-mg once-daily group, 1.15±3.70 in the evobrutinib 75-mg twice-daily group, and 4.78±22.05 in the DMF group. The baseline adjusted rate ratios for the total number of lesions over time as compared with placebo were 1.45 in the evobrutinib 25-mg group (P = 0.32), 0.30 in the evobrutinib 75-mg once-daily group (P = 0.005), and 0.44 in the evobrutinib 75-mg twice-daily group (P = 0.06). The unadjusted annualized relapse rate at week 24 was 0.37 in the placebo group, 0.57 in the evobrutinib 25-mg group, 0.13 in the evobrutinib 75-mg once-daily group, 0.08 in the evobrutinib 75-mg twice-daily group, and 0.20 in the DMF group. There was no significant effect of trial group on the change from baseline in the EDSS score. Elevations in liver aminotransferase values were observed with evobrutinib.
Conclusions: Patients with relapsing multiple sclerosis who received 75 mg of evobrutinib once daily had significantly fewer enhancing lesions during weeks 12 through 24 than those who received placebo. There was no significant difference with placebo for either the 25-mg once-daily or 75-mg twice-daily dose of evobrutinib, nor in the annualized relapse rate or disability progression at any dose. Longer and larger trials are required to determine the effect and risks of evobrutinib in patients with multiple sclerosis. (Funded by EMD Serono; ClinicalTrials.gov number, NCT02975349.).
Paper 2
Background: Tolebrutinib is an oral, CNS-penetrant, irreversible inhibitor of Bruton's tyrosine kinase, an enzyme expressed in B lymphocytes and myeloid cells including microglia, which are major drivers of inflammation in multiple sclerosis. We aimed to determine the dose-response relationship between tolebrutinib and the reduction in new active brain MRI lesions in patients with relapsing multiple sclerosis.
Methods: We did a 16-week, phase 2b, randomised, double-blind, placebo-controlled, crossover, dose-finding trial at 40 centres (academic sites, specialty clinics, and general neurology centres) in ten countries in Europe and North America. Eligible participants were adults aged 18-55 years with diagnosed relapsing multiple sclerosis (either relapsing-remitting or relapsing secondary progressive multiple sclerosis), and one or more of the following criteria: at least one relapse within the previous year, at least two relapses within the previous 2 years, or at least one active gadolinium-enhancing brain lesion in the 6 months before screening. Exclusion criteria included a diagnosis of primary progressive multiple sclerosis or a diagnosis of secondary progressive multiple sclerosis without relapse. We used a two-step randomisation process to randomly assign eligible participants (1:1) to two cohorts, then further randomly assign participants in each cohort (1:1:1:1) to four tolebrutinib dose groups (5, 15, 30, and 60 mg administered once daily as an oral tablet). Cohort 1 received tolebrutinib for 12 weeks, then matched placebo (ie, identical looking tablets) for 4 weeks; cohort 2 received 4 weeks of placebo followed by 12 weeks of tolebrutinib. Participants and investigators were masked for dose and tolebrutinib-placebo administration sequence; investigators, study team members, and study participants did not have access to unmasked data. MRI scans were done at screening and every 4 weeks over 16 weeks. The primary efficacy endpoint was the number of new gadolinium-enhancing lesions detected on the scan done after 12 weeks of tolebrutinib treatment (assessed at week 12 for cohort 1 and week 16 for cohort 2), relative to the scan done 4 weeks previously, and compared with the lesions accumulated during 4 weeks of placebo run-in period in cohort 2. Efficacy data were analysed in a modified intention-to-treat population, using a two-step multiple comparison procedure with modelling analysis. Safety was assessed for all participants who received at least one dose of study drug. This trial is registered with ClinicalTrials.gov (NCT03889639), EudraCT (2018-003927-12), and WHO (U1111-1220-0572), and has been completed.
Findings: Between May 14, 2019, and Jan 2, 2020, we enrolled and randomly assigned 130 participants to tolebrutinib: 33 to 5 mg, 32 to 15 mg, 33 to 30 mg, and 32 to 60 mg. 129 (99%) completed the treatment regimen and 126 were included in the primary analysis. At treatment week 12, there was a dose-dependent reduction in the number of new gadolinium-enhancing lesions (mean [SD] lesions per patient: placebo, 1·03 [2·50]; 5 mg, 1·39 [3·20]; 15 mg, 0·77 [1·48]; 30 mg, 0·76 [3·31]; 60 mg, 0·13 [0·43]; p=0·03). One serious adverse event was reported (one patient in the 60 mg group was admitted to hospital because of a multiple sclerosis relapse). The most common non-serious adverse event during tolebrutinib treatment was headache (in one [3%] of 33 in the 5 mg group; three [9%] of 32 in the 15 mg group; one [3%] of 33 in the 30 mg group; and four [13%] of 32 in the 60 mg group). No safety-related discontinuations or treatment-related deaths occurred.
Interpretation: 12 weeks of tolebrutinib treatment led to a dose-dependent reduction in new gadolinium-enhancing lesions, the 60 mg dose being the most efficacious, and the drug was well tolerated. Reduction of acute inflammation, combined with the potential to modulate the immune response within the CNS, provides a scientific rationale to pursue phase 3 clinical trials of tolebrutinib in patients with relapsing and progressive forms of multiple sclerosis.
Subscriptions and donations
Paid subscriptions to MS-Selfie are being used to administer the Newsletter and associated MS-Selfie microsite that is currently in development. At the request of several readers, I have now added the option of making a one-off donation. To keep this initiative open to all readers, I would appreciate it if those who can afford a subscription please subscribe. For active paying subscribers, thank you; your contribution is much appreciated.
General Disclaimer: Please note that the opinions expressed here are those of Professor Giovannoni and do not necessarily reflect the positions of Barts and The London School of Medicine and Dentistry or Barts Health NHS Trust. The advice is intended as general advice and should not be interpreted as personal clinical advice. If you have problems please tell your own healthcare professional who will be able to help you.



Would an even greater chance of long term success or halting progress be a BTK + an ebv antivirals eg ATA188
Working in conjunction?
Based on a lot of estimates I'm hearing <5 years to market for the BTK. Would you think that's likely or too optimistic?
And is anyone doing a big comparative vs Tysabri / ocrevus / lemtrada etc?
Thank you, Prof. G.
On a different topic, do you recommend that pwMS on Ocrevus who have had a decent response to the vaccine receive Evusheld? Do you have any concerns about risks (e.g., myocarditis) that may be significant but not yet identified because Evusheld has so far been given only to a small number of people (cf. the FDA’s information sheet on Evusheld). Neurologists in the US are starting to recommend Evusheld to their MS patients.